Addressing the Underlying Cardiovascular Hemodynamic Physiology of Upper-Body Stretching in Healthy Adults
Arvind Raj
Webber Academy
Grade 11
Presentation
Problem
Project Introduction Syncope (or “fainting”) is a transient loss of consciousness due to global cerebral hypoperfusion, characterized by a rapid onset, short duration, and spontaneous complete recovery [1-2]. The most common type of syncope is reflex syncope, accounting for 21% of cases [1,3]. Reflex syncope is caused by a “reflex” response in the nervous system, resulting in a large drop in blood pressure (BP) that deprives the brain of adequate blood supply [2,4]. Two main mechanisms cause reflex syncope: the first is vasodepression, where there is inadequate vasoconstriction of the sympathetic nervous system. The second is increased cardioinhibition, where there is increased parasympathetic nervous system (the “yang” to the sympathetic “ying”) drive, causing low heart rate or temporary cessation of heart beating [1,5-6].
There are 4 categories of reflex syncope: vasovagal syncope (VVS), situational syncope, carotid sinus syndrome, and non-classical [1-2]. Situational syncope is an induction of syncope, triggered by a specific emotional, environmental, or physiologic event [4,7]. There are numerous recorded triggers of situational syncope, such as micturition, defecation, swallowing, coughing, and laughing [4,7].
Stretch-induced syncope (SIS) is a rare and poorly understood type of situational syncope, triggered by a “stretching” manoeuvre in affected patients [8-9]. The direct cause of SIS has been subject to limited investigation. Proposed possible causes for SIS include vertebral artery compression, carotid baroreceptor activation, and straining such as in the Valsalva manoeuvre [8,10-11]. These theories are unsubstantiated, however, as a direct result of the lack of literature surrounding SIS [10]. The most recent and comprehensive research on SIS has shown evidence that SIS is triggered by muscle mechanoreceptor activation with hyperextension in upper back and shoulder muscles, resulting in transient, significant hypotension in affected individuals. Further study of the basic reproducibility, symptomology, and treatment of SIS is required [10].
While there are some insights into hemodynamic changes that occur in patients with SIS, it is not known if these changes are pathological or if they are physiological. If they are pathological, then they may affect only the few patients with SIS. Conversely, if they are physiological and are present in control participants without syncope, then this has potentially broader implications for the general population.
Given the current limited understanding of the physiological mechanisms of SIS, this study will use a specialized stretch in a relatively large group of healthy (non-syncopal) adult control participants. This study has the advantage of a large and globally recognized clinic specializing in the clinical care and treatment of patients with syncope, and a state-of-the-art modern human cardiovascular physiology laboratory. We aim to advance the field using state of the art equipment including continuous monitoring of systolic blood pressure (SBP), diastolic blood pressure (DBP), and mean arterial pressure (MAP). This study contributes to determining if the hemodynamic changes seen in SIS are physiological and present in control participants, and provide a better understanding the conditions that can exaggerate these physiological perturbations. Such knowledge helps us better understand the hemodynamic consequences of upper extremity stretch, and circumstances to minimize these hemodynamic consequences and potentially minimize symptoms.
Research Questions
- Does upper body stretch cause a significant BP drop in healthy (without syncope) volunteers?
- Does the duration of upper body stretch influence the induced hypotension?
- Does orthostatic stress increase the induced hypotension with upper body stress?
Hypotheses
- Upper body stretching causes a significant drop in BP in healthy (without syncope) volunteers.
- A longer upper body stretch will result in a larger BP drop in healthy (without syncope) volunteers than a shorter upper body stretch.
- An upper body stretch while in the upright posture will result in a larger BP drop in healthy (without syncope) volunteers than a similar upper body stretch while lying down.
Objectives Short-term goals: To analyse data on measured SBP, DBP, and MAP hemodynamic variables during performance of a stretch in healthy (no prior syncope) volunteers in isolation; then to compare a similar stretch of variable durations, and to compare the stretches when lying down and when standing.
Long-term goals: To use the conclusions made about these physiological mechanisms in response to upper body stretch to educate people about how to stretch and minimize the risks of significant, dramatic reductions in BP. Further, the findings gleaned from this study will be used to guide future research into understanding the hemodynamic effects of upper body stretch and SIS. Our findings may also be relevant to helping patients with other types of syncope, as they may share physiological mechanisms according to other studies.
Method
Recruitment and Participant Eligibility Healthy control participants were eligible if they had: no history of syncope, could perform the stretch maneuver, and were between 18 and 60 years of age with informed written consent. All patients were excluded if they had: (1) an inability to give informed consent, (2) important valvular, coronary, myocardial or conduction abnormality or significant arrhythmia, (3) hypertrophic cardiomyopathy, (4) a permanent pacemaker, (5) a seizure disorder, (6) hypertension defined as greater than 160/90 mmHg, (7) pregnancy, (8) were lactating women, (9) medications with known effects on BP, and (10) other factors which, in the investigator’s opinion, would prevent the subject from completing the protocol.
Instrumentation Instrumentation included conventional electrocardiogram (ECG) electrodes to monitor continuous heart rhythm, although collected HR data was unnecessary for this study. BP was monitored continuously beat-to-beat using a Finapres NOVA finger BP device. Prior to stretch, the beat-to-beat BP was calibrated with brachial arm BP cuff measurements. All analogue signals, including SBP, DBP, and MAP, were sampled at >100 Hz (PowerLab, ADInstruments, Colorado Springs, CO, USA) and stored digitally for off-line analysis and data extraction using custom software written in MATLAB r2024b (Mathworks, Natick, MA, USA).
Testing Protocol Participants lay supine for 30 seconds while initial baseline (no stretch) data was collected. Then, participants would perform the stretch maneuver that consists of (1) upper back hyperextension, (2) shoulder abduction of the arm not wearing the fingertip blood pressure monitor, and (3) neck hyperextension. The position was held for 15 seconds. This maneuver was repeated 30 seconds later; participants would lie supine quietly for a duration of 30 seconds while repeating the maneuver. We would then actively stand participants up and then repeat the stretch for 15 seconds and then 30 seconds while upright. A 30 second baseline was performed before each stretch. Continuous data are presented as mean±standard deviation. Categorical data are presented as number (%). Analysis of the blood pressure drop from baseline with upper body stretch was performed with a one-group t-test [17]. Comparisons of 15 second vs. 30 second stands (question #2) and upper body stretch while lying down vs. while standing up (question #3) were analysed using paired t-tests [17]. Paired analyses were repeated with the non-parametric Wilcoxon Signed Rank test [18]. All paired analyses had qualitatively similar results with the parametric and non-parametric analyses. A 2-tailed p-value of ≤0.05 was deemed to be statistically significant. Statistical analyses were performed using SPSS statistical software for Windows version 29 (SPSS, Inc., IBM, Armonk, NY). Figures were created using GraphPad Prism (version 8.0.0 for Windows, GraphPad Software, San Diego, California USA, www.graphpad.com). There was no specific data with which to base sample size estimates. We proposed a sample size of up to 20 healthy adult participants; we gathered and statistically analyzed data on 17 healthy adult participants, although participant enrolment is currently ongoing. This methodology and project were approved by the Calgary Conjoint Health Research Ethics Board (REB15-3080).
Research
One study found that cardiovascular responses during neck hyperextension were similar to those seen during a Valsalva manoeuvre [12], including increased heart rate during straining. This did not occur when the neck was straight. It was clearly noted, however, that other clinical features that suggested additional mechanisms may be involved for induced syncope. For example, visual auras were common, which are associated with posterior brain circulation impairment [8].
A physiologic study was performed on a population of 2 SIS patients, with various stretched positions and use of the Valsalva manoeuvre, while using Transcranial Doppler of the posterior cerebral arteries and angiography to examine brain blood flow in real time. Maximal head extension and arm hyperabduction (arms stretched away from the midline) produced decreased posterior cerebral artery blood flow. Dynamic angiography showed extracranial compression of the craniocervical arteries in both patients, supporting direct vertebral artery compression during stretching. It was concluded that hyperextension of the head and neck is needed to induce SIS, meaning that the Valsalva manoeuvre (which occurs when stretching, but not specifically the neck) alone does not cause SIS. Decreased blood flow to the posterior cerebral circulation must therefore be an important contributing factor to SIS [9].
Three male adolescents with SIS were studied using continuous video electroencephalogram (EEG), transcranial Doppler of the middle cerebral artery (MCA), and continuous BP monitoring. All episodes started with a period of stretching and breath holding, then impaired consciousness was seen 9-16 secs later. The stretch was associated with increase heart rate and reduced BP, along with cessation of MCA diastolic blood flow when the BP dropped. EEG showed generalized slow waves consistent with impaired consciousness. As slow wave EEG abnormalities reflect generalized cerebral dysfunction, it was concluded that the delay between stretching and the occurrence of low BP and EEG changes suggests that a reflex mechanism is likely in SIS. SIS is therefore a type of vasodepressor (reflex) syncope, pointing away from it being caused by vertebrobasilar compression alone [13].
Transcranial colour-coded duplex sonography in an 8-year-old girl with SIS showed reversal of blood flow in the right vertebral artery with the head turned to the left at 45 degrees, and the patient felt faint. This reversed when the head was placed in a neutral position. This suggests that reduced blood flow in the posterior circulation of the brain (as shown by the reversal of blood flow in the vertebral artery on neck extension) likely contributed to the syncope in this patient (also known as vertebrobasilar insufficiency). Furthermore, the local and generalized EEG slowing observed suggested reduced cerebral blood supply due to a drop in BP [14]. A study in a 7-year-old girl with SIS showed dynamic compression of the vertebral arteries induced a reflex syncope (blood vessel dilation and low heart rate), causing a transient decrease in blood flow to the brainstem [14-15].
Recently, physiologic studies were performed on 9 patients with SIS and 24 healthy controls. Subjects performed the active stand test, Valsalva manoeuvre, respiratory sinus arrhythmia, head-up tilt test, and active shoulder/upper back extension manoeuvre (shrug). They monitored heart rate (HR), systolic blood pressure (SBP, pressure of blood when the heart contracts), and mean arterial pressure (MAP, average pressure in a person’s arteries, during one cardiac cycle [average of diastolic {DBP} and systolic blood pressure, during a cardiac cycle]). During stretching, 7/9 suspected SIS patients (78%) either developed syncope (2 patients) or pre-syncope (5 patients). In the control group, 5 patients experienced light-headedness and a concomitant drop in BP, but none experienced syncope or pre-syncope. They found that SIS symptoms may be reproduced (in SIS patients) when transient systemic hypotension is triggered through the hyperextension of upper back and shoulder muscles, relating to the activation of muscle mechanoreceptors in these areas. It was concluded that SIS is likely the result of a neural reflex mechanism and is therefore not a result of a previously proposed Valsalva response or vertebral artery constriction [10].
Data
Table 1. Participant demographics
| Parameter (units) | Mean±SD |
|---|---|
| Demographics (n=17) | |
| Age (years) | 38 ± 17 |
| Female (n) | 15 (88%) |
| Height (cm) | 168 ± 7 |
| Weight (kg) | 70 ± 21 |
| BMI (kg/m2) | 24.9 ± 8.0 |
| BSA (m2) | 1.79 ± 0.25 |
Abbr: BMI, Body Mass Index; BSA, Body Surface Area.
Demographics Seventeen healthy control participants without a history of syncope participated in the protocol (F=15; age 38±17 years). Participant demographics are provided in Table 1. In order to perform statistical analysis on participant data, seventeen participants were involved in results analysis, rather than the final expected study population of twenty.
Table 2. Parameters measured at nadir systolic blood pressure during upper body stretch (Question #1).
| Parameter (units) | Mean±SD | P Value |
|---|---|---|
| Lying Down – 15 second Stretch | ||
| SBP (mmHg) | -13.3 ± 7.7 | <0.001 |
| DBP (mmHg) | -7.6 ± 10.0 | 0.006 |
| MAP (mmHg) | -9.5 ± 8.4 | <0.001 |
| Lying Down – 30 second Stretch | ||
| SBP (mmHg) | -14.9 ± 11.2 | <0.001 |
| DBP (mmHg) | -6.8 ± 5.3 | <0.001 |
| MAP (mmHg) | -9.7 ± 6.6 | <0.001 |
| Standing Up – 15 second Stretch | ||
| SBP (mmHg) | -25.6 ± 19.1 | <0.001 |
| DBP (mmHg) | -16.4 ± 9.8 | <0.001 |
| MAP (mmHg) | -19.4 ± 12.3 | <0.001 |
| Standing Up – 30 second Stretch | ||
| SBP (mmHg) | -19.8 ± 10.8 | <0.001 |
| DBP (mmHg) | -14.6 ± 7.3 | <0.001 |
| MAP (mmHg) | -16.5 ± 8.4 | <0.001 |
Reported P values are from one-group t-tests compared to no change with stretch (0 mmHg). Abbr: SBP, systolic blood pressure; DBP, diastolic blood pressure; MAP, mean arterial pressure
Question #1: Does upper body stretch cause a significant drop in blood pressure?
A 15-second upper body stretch performed while lying down caused a significant decrease in systolic blood pressure (-13.3 ± 7.7 mmHg; P<0.001), diastolic blood pressure (-7.6 ± 10.0 mmHg; P=0.006), and mean arterial pressure (-9.5 ± 8.4 mmHg; P<0.001). Data are shown in Table 2. There were significant blood pressure drops seen with a 15-second upper body stretch performed while standing up, and 30-second upper body stretches performed while lying down and while standing up (Table 2).
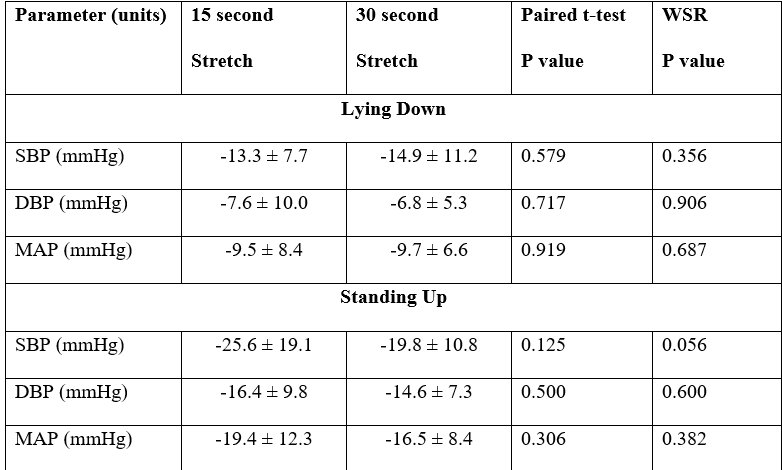
Table 3. Comparison of drops in blood pressure at nadir systolic blood pressure between regular upper body stretch (15 seconds) compared to prolonged upper body stretch (30 seconds) (Question #2).
 Reported P values are from the parametric paired t-tests and the non-parametric Wilcoxon Signed Rank test. Abbr: WSR, Wilcoxon Signed Rank test; SBP, systolic blood pressure; DBP, diastolic blood pressure; MAP, mean arterial pressure
Reported P values are from the parametric paired t-tests and the non-parametric Wilcoxon Signed Rank test. Abbr: WSR, Wilcoxon Signed Rank test; SBP, systolic blood pressure; DBP, diastolic blood pressure; MAP, mean arterial pressure
Question #2: Does a longer duration of upper body stretch (30 seconds) cause a greater drop in blood pressure compared to a nominal stretch (15 seconds)?
There were no differences between the magnitude of hypotension induced by upper body stretch lasting 15 seconds or 30 seconds, whether they were performed lying down or standing up (Table 3).
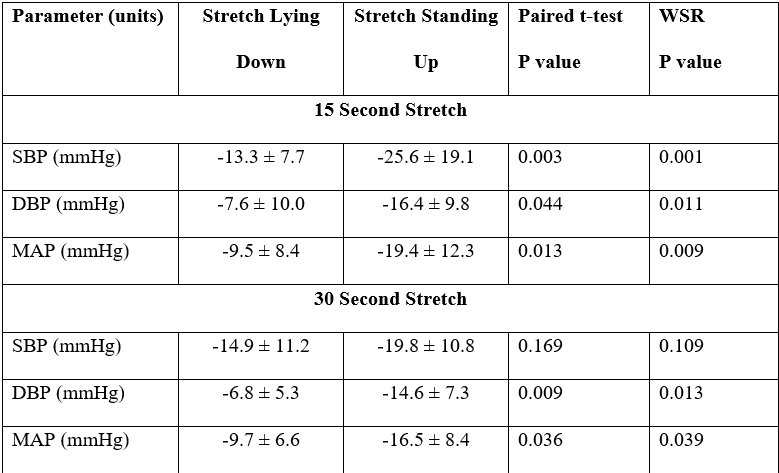
Table 4. Comparison of drops in blood pressure at nadir systolic blood pressure between upper body stretch while lying down compared to upper body stretch while standing up (Question #3).
 Reported P values are from the parametric paired t-tests and the non-parametric Wilcoxon Signed Rank test. Abbr: WSR, Wilcoxon Signed Rank test; SBP, systolic blood pressure; DBP, diastolic blood pressure; MAP, mean arterial pressure
Reported P values are from the parametric paired t-tests and the non-parametric Wilcoxon Signed Rank test. Abbr: WSR, Wilcoxon Signed Rank test; SBP, systolic blood pressure; DBP, diastolic blood pressure; MAP, mean arterial pressure
Question #3: Does upper body stretch during upright posture cause a greater drop in blood pressure compared to upper body stretch while lying down?
Upper body stretch for 15 seconds while upright, compared to while lying down, induced a greater drop in systolic blood pressure (-25.6 ± 19.1 mmHg vs. -13.3 ± 7.7 mmHg; P=0.001), diastolic blood pressure (-16.4 ± 9.8 mmHg vs. -7.6 ± 10.0 mmHg; P=0.011), and mean arterial pressure (-19.4 ± 12.3 mmHg vs. -9.5 ± 8.4 mmHg; P=0.009). When the upper body stretch was extended to 30 seconds, upright posture induced a significantly greater drop in blood pressure than when lying down for diastolic blood pressure (-14.6 ± 7.3 mmHg vs. -6.8 ± 5.3 mmHg; P=0.013) and mean arterial pressure (-16.5 ± 8.4 mmHg vs. -9.7 ± 6.6 mmHg; P=0.039), with a non-statistically significant trend toward a greater drop in systolic blood pressure (-19.8 ± 10.8 mmHg vs. -14.9 ± 11.2 mmHg; P=0.109).
Conclusion
Limitations The sample size of 17 participants is a fairly good size for a physiological study, but it is still modest. A larger sample size might have resulted in a statistically significant difference in the magnitude of the drop in systolic blood pressure with a prolonged upper body stretch when upright compared to when lying down. We plan to continue enrolment, in order to negate this limitation. Almost 90% of our participants were female. This limits our ability to extrapolate our findings & conclusions to males, over different sex-based physiologies. We studied participants between the ages of 18-60 years. We cannot comment on the effect of upper body stretch-induced hypotension in children and in older adults, which limits the applicability of our research to some degree. Conclusions In this study, we were able to show that upper body stretch induced significant hypotension in healthy participants. While we hypothesized that prolonging the duration of stretch would increase the magnitude of the induced hypotension, our hypothesis was wrong. Prolonging the duration of stretch did not alter the amount of induced hypotension. Finally, we found that upper body stretch induced greater hypotension when performed in the upright body position than when lying down in healthy (non-syncopal) participants. These data show that upper body stretch induced hypotension is a physiological phenomenon, and not a pathology unique to stretch-induced syncope. Rather, patients with stretch-induced syncope likely represent an extreme of another otherwise normal physiological phenomenon. These findings also have implications for the average person with upper body stretch-induced hypotension. First, it is important for people to realize that lightheadedness that occurs during or just after stretching could be explicitly due to the stretching; this suggests that stretching could be a significant potential trigger for pre-syncopal or syncopal induction, as with other well-documented multifactorial syncopal triggers, such as dehydration and high body temperature [19-20]. Second, and perhaps more importantly, if someone is prone to lightheadedness with their upper body stretch-induced hypotension, then they should be advised to perform their upper body stretches while lying down as opposed to after standing up. Additionally, upper body stretches under the condition of standing with a 15 second stretch (U15) produced a mean drop in BP of about 25.6 mmHg with a high standard deviation (±19.1 mmHg). This suggests that some individuals, specifically under the condition of orthostatic stress, had BP drops of greater than 40 mmHg during stretch. At this degree of induced hypotension, individuals have been shown to be prone to spells of lightheadedness, pre-syncope, or even syncopal induction; these symptoms mirror those caused by SIS, suggesting that SIS may be more common than is currently believed (with this study only examining a sample of n=17). Future Directions Going forward, we plan to first finish the planned enrolment in this study. We have a target sample size of 20. We will plan to study younger and older individuals to determine the impact of upper body stretch induced hypotension across the lifespan. Finally, we plan to additionally enrol study patients with stretch-induced hypotension (SIS individuals) to determine if these patients have a greater magnitude of hypotension than our healthy (non-syncope) participants.
Citations
References 1. Romano S\, Branz L\, Fondrieschi L\, Minuz P. Does A therapy for reflex vasovagal syncope really exist? High Blood Press Cardiovasc Prev. 2019;26(4):273-281. doi:10.1007/s40292-019-00327-3 2. Brignole M\, Moya A\, de Lange FJ\, et al. Practical Instructions for the 2018 ESC Guidelines for the diagnosis and management of syncope. Eur Heart J. 2018;39(21):e43-e80. doi:10.1093/eurheartj/ehy071 3. Soteriades ES\, Evans JC\, Larson MG. Incidence and prognosis of syncope. ACC Curr J Rev. 2003;12(1):74. doi:10.1016/s1062-1458(02)01060-7 4. Johansson M\, Fedorowski A. Situational vs vasovagal syncope: one but different? Heart. 2023;110(1):3-4. doi:10.1136/heartjnl-2023-323180 5. Mosqueda-Garcia R\, Furlan R\, Tank J\, Fernandez-Violante R. The elusive pathophysiology of neurally mediated syncope. Circulation. 2000;102(23):2898-2906. doi:10.1161/01.cir.102.23.2898 6. Morillo CA\, Eckberg DL\, Ellenbogen KA\, et al. Vagal and sympa- thetic mechanisms in patients with orthostatic vasovagal syncope. Circulation. 1997;96:2509-2513. 7. Zou R\, Wang S\, Lin P\, et al. The clinical characteristics of situational syncope in children and adults undergoing head-up tilt testing. Am J Emerg Med. 2020;38(7):1419-1423. doi:10.1016/j.ajem.2019.11.042 8. Pelekanos JT\, Dooley JM\, Camfield PR\, Finley J. Stretch syncope in adolescence. Neurology. 1990;40(4):705-707. doi:10.1212/wnl.40.4.705 9. Sturzenegger M\, Newell DW\, Douville CM\, Byrd S\, Schoonover KD\, Nicholls SC. Transcranial Doppler and angiographic findings in adolescent stretch syncope. J Neurol Neurosurg Psychiatry. 1995;58(3):367-370. doi:10.1136/jnnp.58.3.367 10. Kosmopoulos M\, Reyes JL\, Patel S\, et al. Stretch syncope in humans: Evidence for symptomatic neural reflex hypotension triggered by stretching of shoulder and upper back muscles. Heart Rhythm. 2025;22(6):1464-1471. doi:10.1016/j.hrthm.2025.03.166 11. Mazzuca M\, Thomas P. Self-induced stretch syncope of adolescence: a video- EEG documentation. Epileptic Disord. 2007;9:413-417. 12. Hiner BC. Valsalva maneuver. Clin Med Res. 2005;3(2):55; author reply 55. doi:10.3121/cmr.3.2.55 13. Sarrigiannis PG\, Randall M\, Kandler RH\, Grunewald RA\, Harkness K\, Reuber M. Stretch syncope: reflex vasodepressor faints easily mistaken for epilepsy. Epilepsy Behav. 2011;20(3):450-453. doi:10.1016/j.yebeh.2010.12.013 14. Mercante A, Pizza F, Pondrelli F, et al. Self-induced stretch syncope: An unusual non-epileptic paroxysmal event. A case report and literature mini-review. Clin Med Insights Pediatr. 2024;18:11795565241249596. doi:10.1177/11795565241249596 15. Routier L\, Bourel-Ponchel E\, Heberle C\, et al. Stretch syncope or epileptic seizure? A pathologic hypothesis for self-induced stretch syncope. Neurophysiol Clin. 2020;50(5):383-386. doi:10.1016/j.neucli.2020.08.002 16. Barritt AW\, MacDonald BK. Breakfast time blackouts. Case Rep Neurol Med. 2014;2014:568983. doi:10.1155/2014/568983 17. Thukral S\, Kovac S\, Paturu M. T-test. In: Translational Interventional Radiology. Elsevier; 2023:139-143. doi:10.1016/b978-0-12-823026-8.00104-8 18. Understanding the Wilcoxon Signed Rank Test. Statistics Solutions. December 21\, 2010. Accessed March 1\, 2026. https://www.statisticssolutions.com/free-resources/directory-of-statistical-analyses/how-to-conduct-the-wilcox-sign-test/ 19. Brignole M. Diagnosis and treatment of syncope. Heart. 2007;93(1):130-136. doi:10.1136/hrt.2005.080713 20. Syncope (Fainting). www.heart.org. Accessed March 1\, 2026. https://www.heart.org/en/health-topics/arrhythmia/symptoms-diagnosis--monitoring-of-arrhythmia/syncope-fainting 21. Bruss ZS\, Raja A. Physiology\, stroke volume. In: StatPearls. StatPearls Publishing; 2025. https://www.ncbi.nlm.nih.gov/pubmed/31613466 22. Trammel JE\, Sapra A. Physiology\, systemic vascular resistance. In: StatPearls. StatPearls Publishing; 2025. https://www.ncbi.nlm.nih.gov/pubmed/32310535 23. Vincent JL. Understanding cardiac output. Crit Care. 2008;12(4):174. doi:10.1186/cc6975 24. King J\, Lowery DR. Physiology\, cardiac output. In: StatPearls. StatPearls Publishing; 2025. https://www.ncbi.nlm.nih.gov/pubmed/29262215 25. Ringer M\, Hashmi MF\, Lappin SL. Orthostatic hypotension. In: StatPearls. StatPearls Publishing; 2025. Accessed December 19\, 2026. https://www.ncbi.nlm.nih.gov/books/NBK448192/ 26. McHugh ML. The chi-square test of independence. Biochem Med (Zagreb). 2013;23(2):143-149. doi:10.11613/bm.2013.018
Image Links: 1. https://www.istockphoto.com/photos/ecg-electrodes (Electrocardiogram Electrodes) 2. https://www.medtach.com/finapres-continuous-blood-pressure.html (Finapres NOVA Monitor) 3. https://en.ac-illust.com/clip-art/1964671/stickman-stretch (Person Stretching Clipart) 4. https://www.istockphoto.com/illustrations/green-check-mark 5. https://gallery.yopriceville.com/Free-Clipart-Pictures/Arrows-PNG/Check_Marks_PNG_Clipart 6. https://www.studentquickpay.com/webberacademy/ (Webber Academy Logo) 7. https://www.slps.ca/university-of-calgary (University of Calgary Logo) 8. https://libin.ucalgary.ca/about-us/our-brand (Libin Cardiovascular Institute Logo) 9. https://www.news-medical.net/news/20251207/Study-reveals-the-hidden-toll-of-sudden-cardiac-death-in-type-1-and-type-2- diabetes.aspx (Heart Beating Image\, Front Page of Logbook & Project Banner Image)
Acknowledgement
I would first like to acknowledge my Science Fair Coordinator and (one of two) Applied Science Project (ASP) instructor(s), Dr. Garcia, for providing me ample advice on how to organize my time, efforts, and energy for this Science Fair project, in order to ensure optimal completion and best practice. I would additionally like to thank Dr. Garcia for providing me what felt like as many extensions as I could ask for, in order to complete my ASP assignments on time, while still balancing my heavy Science Fair workload.
I would next like to acknowledge Mrs. Kale, my second Applied Science Project Instructor, for regularly meeting with me and helping me to ensure that I am making progress on my project, and that I was not just leaving it to the wayside under the pressures of the ASP course.
I would like to acknowledge my primary mentor, Dr. Robert S. Sheldon, for giving me guidance and advice throughout the course of the project, helping me find new direction when the course of my project had hitches and turns, and offering me his expertise whenever I had a written project for ASP that I was uncertain the quality of. Additionally, I would really like to emphasize just how thankful I am to Dr. Sheldon for affording me his laboratory resources and involving me in this research journey, to begin with.
I would like to acknowledge Rashmin Hira, a PhD candidate working with Dr. Sheldon in the Autonomic Clinic, who ran Autonomic Function Test (AFT) trials while I was not available to collect data myself (September onward). I would additionally like to acknowledge Rashmin for helping me access any necessary demographic or stretch data that I was not able to get my hands on, personally.
I would like to acknowledge Priyanshu Giri, an undergraduate student working with Dr. Sheldon (majoring in Biology) who taught me how to use SPSS, aided significantly with data collection, was the first line of review for any documents that I wanted to send to Dr. Sheldon, and provided me crucial workflow and productivity advice throughout the course of my project.
I would like to acknowledge Martin Miranda Hurtado, a MATLAB-experienced coder who works with Dr. Sheldon, Rashmin, and Priyanshu, for creating a MATLAB code, tailored to my specifications, that allowed me to extract data from signal-processed stretch files.
I would like to acknowledge Rasha Hamzeh, the Laboratory Manager working alongside Dr. Sheldon, for providing me pertinent lab documents and protocols wherever & whenever necessary.
Note: AI was not used for this project. All parts of the project report were hand-written, data was extracted using a MATLAB-based code, and all project information was gathered manually (with some minor guidance from Dr. Sheldon). PaperPile, an external reference manager provided to ASP students, was used for resource access-dates and proper AMA citation formatting.

