Cytokine Balance, Pneumonia, and the Impact of Past Influenza or COVID-19 Infections
Avneet Gill
STEM Innovation Academy Jr. High N.W. Campus
Grade 9
Presentation
No video provided
Problem
Pneumonia is an infection of the lungs that can cause mild illness in some people, but severe lung damage or even death in others. The difference in these outcomes does not depend solely on the bacteria or virus causing the infection, suggesting that additional factors influence how pneumonia affects the body. One critical factor is how the immune system responds, particularly through chemical signals called cytokines (National Heart, Lung, and Blood Institute; PubMed review).
Inflammation is the body’s natural defense response to infection and injury. It causes blood vessels to widen and become more permeable, allowing immune cells to reach the infected tissue. Cytokines act as signaling molecules that control this process by instructing immune cells when to activate, where to move, and how strong the response needs to be. Because inflammation can both protect and damage the body, cytokine signaling must be carefully regulated. If this regulation becomes unbalanced, inflammation may become excessive or insufficient, increasing the risk of tissue damage or delaying recovery (PubMed review). Cytokines regulate inflammation so that it is neither too high nor too low, maintaining balance-an essential part of an effective immune response. This regulation helps immune cells reach the lungs and fight infection efficiently. Pro-inflammatory cytokines aid in eliminating pathogens, but if their activity is excessive, they can damage lung tissue and fill the alveoli with fluid, reducing oxygen exchange and potentially leading to severe pneumonia, breathing difficulties, or respiratory failure. Anti-inflammatory cytokines reduce inflammation and promote healing, but if they act too early or too strongly, they can slow infection clearance. The challenge for the immune system is to maintain balance; when this balance is lost, inflammation can shift from being protective to harmful (PubMed review; National Heart, Lung, and Blood Institute).
Additionally, many individuals experience respiratory viral infections such as influenza or COVID-19 prior to developing pneumonia. It is not yet fully understood how these previous infections influence cytokine regulation during later pneumonia episodes (PubMed review).
The problem addressed in this project is the lack of understanding of how cytokine balance influences pneumonia outcomes, and whether a history of viral respiratory infection is associated with differences in inflammatory responses during pneumonia (PubMed review; National Heart, Lung, and Blood Institute).
Method
Abstract
This project is about how cytokines, which are chemicals in the immune system, affect pneumonia. It also looks at whether or not having viruses in the past, like influenza or COVID-19, can change how the immune system reacts. This project uses information from studies that already exist, including medical studies, research articles, and reviews that are available online. Using already published research makes this project possible and reliable. The information is collected and organized to help answer the research question and show how scientific data can still be used in a school project.
1. Immune System Factors
The immune system has many parts that work together to fight infections. Cytokines are one of the main ways immune cells communicate with each other. They control how strong inflammation is during an infection like pneumonia.
Some cytokines increase inflammation. These are called pro-inflammatory cytokines, and examples include IL-6 and TNF-a. They help the body fight infection, but if there are too many of them, they can damage lung tissue. Other cytokines reduce inflammation. These are anti-inflammatory cytokines, such as IL-10. While they help calm the immune system, too much of this response can slow down the removal of bacteria or viruses.
Looking at both types of cytokines helps explain why balance is important in pneumonia. Cytokines were chosen because many studies already measure them, so the data is easier to find.
2. Sources and Data Collection
2.1 Sources
The sources for this project come from online databases like PubMed and Google Scholar. Only studies that were related to my topic, and provided me with relevant information for this project in the abstract were chosen. Preference was given to studies that included:
- Cytokine data
- Patients with pneumonia
- Information about past viral infections when available
- Clear explanations of how the study was done
Most sources are peer-reviewed to make sure the information is reliable.
2.2 Data Collection
Data was taken directly from studies like PubMed or articles on Google Scholar. This included cytokine levels, patient age, and how severe the pneumonia the patient had was. If the study mentioned any past infections or diseases, that information is also included.
The data was organized into tables/graphs to make comparisons easier to interpret. Each study is clearly labeled with its title and year so the information could be checked again if needed.
3. Organizing and Comparing Data
3.1 Grouping
After the data was collected, it was grouped to make comparisons clearer. Groups included mild, moderate, and severe pneumonia. Other groups were based on age and whether patients had past viral infections. Therefore, helping reduce confusion, and making the data and patterns easy to understand.
3.2 Comparison
The data in each group was compared to look for trends. For example, higher cytokine levels in severe cases compared to mild cases. Simple tables or graphs can be used to show differences. The goal is to observe patterns, not to prove cause and effect. Ultimately, leading this project to a fair conclusion.
4. Reliability and Limits
To improve reliability, more than one study was used. If similar results appeared in different studies, they were more likely to be accurate. All data was taken from trusted sources and carefully recorded in the citations section and furthermore in my reference document.
There are limits to this type of research. Studies do not always measure cytokines the same way, and not all studies include information about past viral infections. This can make comparisons harder.
Research
Brief Introduction to the Topic
Pneumonia is a serious respiratory illness that develops when the lungs become infected by bacteria, viruses, or, in some cases, fungi. The infection primarily affects the alveoli, which are tiny air sacs responsible for the exchange of oxygen and carbon dioxide. During pneumonia, these air sacs become inflamed and may fill with fluid or pus, limiting the movement of oxygen into the bloodstream and producing symptoms such as coughing, fever, chest pain, and difficulty breathing (National Heart, Lung, and Blood Institute). In severe cases, impaired gas exchange can lead to low oxygen levels and increased strain on vital organs (pathophysiology overview). Pneumonia poses a particularly high risk to young children, older adults, and individuals with weakened immune systems, making it a major public health concern worldwide (National Heart, Lung, and Blood Institute).
Inflammation plays a central role in the development and progression of pneumonia and represents a natural defense mechanism of the immune system. When pathogens enter the lungs, immune cells trigger a localized inflammatory response that increases blood flow to the infected area and helps recruit white blood cells and protective proteins to contain and eliminate the infection (pathophysiology overview). Cytokines are chemical signaling molecules released by immune cells during this response that help coordinate inflammation and immune cell activity (PubMed review). Under controlled conditions, inflammation supports the clearance of pathogens and recovery. Problems arise when inflammation becomes excessive or prolonged, which can increase fluid accumulation within the alveoli, damage healthy lung tissue, and interfere with normal breathing, ultimately worsening symptoms and delaying recovery (pathophysiology overview).
Effective immune responses depend on balance rather than intensity alone. A well-regulated immune response allows the body to eliminate infectious agents while minimizing harm to healthy lung tissue. An overly aggressive response can cause unnecessary lung injury, while an insufficient response may allow pathogens to persist or spread deeper into the lungs (PubMed review). Understanding how cytokine balance influences inflammation provides a foundation for explaining why pneumonia severity varies between individuals and directly supports the research question investigating whether immune regulation promotes recovery or contributes to lung damage.
The Immune Response in the Lungs
When bacteria or viruses enter the lungs, the immune system responds quickly to prevent the infection from spreading deeper into lung tissue. The lungs are constantly exposed to air from the environment, which means they must defend against pathogens while still allowing efficient gas exchange to occur (National Heart, Lung, and Blood Institute). Once a pathogen reaches the alveoli, specialized immune cells recognize it as a threat and trigger an immune response. This response initiates inflammation, increasing blood flow and allowing immune cells and defensive proteins to move into the infected area. The purpose of this process is to eliminate the pathogen while limiting damage to the thin, delicate lung structures that are essential for breathing (PubMed review).
Macrophages are among the first immune cells to respond during pneumonia and play a central role in initiating lung immunity. These cells normally reside in the alveoli, where they constantly monitor inhaled air for harmful particles. When macrophages detect bacteria or viruses, they engulf and break down the pathogen through a process called phagocytosis. Beyond directly removing infectious agents, macrophages release signaling molecules that alert the rest of the immune system to the presence of infection. This signaling step is critical, as it determines how strongly the immune response will develop and which additional immune cells will be recruited to the lungs (PubMed review).
Neutrophils are rapidly recruited to the lungs following these early immune signals. These cells are highly effective at killing pathogens, particularly bacteria, by releasing enzymes and reactive molecules that destroy invading organisms. Their strong antimicrobial activity makes them essential for clearing pneumonia-causing infections. However, neutrophils do not distinguish perfectly between pathogens and healthy tissue. When present in excessive numbers or activated for too long, they can damage alveolar walls and worsen inflammation. This dual role highlights why neutrophil activity must be tightly controlled during pneumonia to ensure effective pathogen clearance without unnecessary lung injury (National Institutes of Health).
Coordination between macrophages, neutrophils, and other immune cells is essential for determining pneumonia outcomes. This coordination is largely controlled through chemical signaling rather than direct cell-to-cell contact. Immune cells rely on signaling molecules to regulate how many cells are recruited, how long inflammation persists, and when the immune response should begin to slow. These signals allow the immune system to adjust its intensity based on the severity of infection. Poor regulation at this stage can result in either excessive inflammation that damages lung tissue or an insufficient response that allows the infection to persist (PubMed review).
Cytokines serve as the primary signaling molecules that regulate this immune coordination in the lungs. Macrophages and neutrophils release cytokines to communicate with each other, controlling the strength and duration of inflammation. These signals act as a bridge between immune cell activity and inflammation control, ensuring that the response remains targeted and time-limited. Understanding how immune cells interact through cytokine signaling provides essential context for examining how immune regulation influences pneumonia severity and prepares the foundation for exploring the role of cytokine balance in later sections (PubMed review).
Figure 1: Diagram illustrating the immune response in the lungs during pneumonia, showing macrophages detecting pathogens in the alveoli, neutrophil recruitment, and cytokine signaling pathways that regulate inflammation and immune coordination.
Cytokines: What they Are, and Why They Matter
Cytokines are small proteins that act as communication signals within the immune system. Rather than directly destroying bacteria or viruses, cytokines coordinate how immune cells behave during an infection. They determine when immune cells activate, where they move, how long they remain active, and when the immune response should begin to slow (PubMed review). In pneumonia, cytokines are especially important because the lungs must respond quickly to infection while protecting the fragile alveoli responsible for oxygen exchange. Without proper cytokine signaling, immune cells cannot work together effectively, increasing the risk of uncontrolled inflammation or persistent infection (National Heart, Lung, and Blood Institute). Cytokines play a central role in controlling inflammation in the lungs. When a pathogen is detected, immune cells such as macrophages release pro-inflammatory cytokines that increase blood vessel permeability and attract additional immune cells to the infected area. This process allows neutrophils and other immune cells to reach the alveoli and eliminate invading organisms. At the same time, other cytokines act to limit inflammation by reducing immune cell activity once the infection begins to resolve. These opposing signals allow the immune system to adjust its response as conditions change, ensuring that inflammation remains effective rather than destructive (PubMed review).
The function of cytokines can be compared to a volume control or thermostat for the immune response. When pneumonia begins, cytokines turn the immune “volume” up so the body can respond strongly enough to control the infection. As pathogens are cleared, different cytokines gradually turn the volume back down to prevent damage to healthy lung tissue. Problems arise when this adjustment fails. If the volume remains too high for too long, inflammation can injure the alveoli and interfere with breathing. If the volume stays too low, the immune system may fail to eliminate the infection completely. This analogy highlights why cytokines are not simply triggers of inflammation but regulators that determine how severe pneumonia becomes (PubMed review).
Cytokine signaling also connects immune cell activity to overall disease progression. Macrophages, neutrophils, and other immune cells rely on cytokines to coordinate their actions rather than acting independently. This signaling ensures that immune responses remain targeted to infected areas and do not spread unnecessarily throughout lung tissue. When cytokine signaling becomes poorly regulated, immune cells may continue releasing inflammatory substances even after the pathogen is controlled. This loss of regulation increases the risk of lung tissue damage and prolonged recovery, emphasizing why cytokines play a decisive role in pneumonia outcomes (PubMed review).
Understanding cytokines provides essential context for examining how pneumonia severity varies between individuals. Differences in cytokine production, timing, and regulation can determine whether inflammation remains protective or becomes harmful. This sets the foundation for exploring cytokine balance, where the interaction between pro-inflammatory and anti-inflammatory signals determines whether the immune response supports recovery or contributes to lung injury (PubMed review).
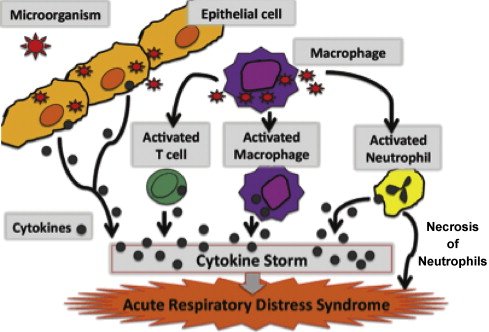 Figure 2: Diagram illustrating cytokine signaling during pneumonia, showing immune cells releasing pro-inflammatory and anti-inflammatory cytokines that regulate inflammation intensity and duration in the lungs.
Figure 2: Diagram illustrating cytokine signaling during pneumonia, showing immune cells releasing pro-inflammatory and anti-inflammatory cytokines that regulate inflammation intensity and duration in the lungs.
Cytokine Balance and Pneumonia Outcomes
The outcome of pneumonia is not determined solely by the presence of bacteria or viruses in the lungs, but by how the immune system regulates inflammation in response to these pathogens. Central to this regulation is the balance between pro-inflammatory and anti-inflammatory cytokines. Pro-inflammatory cytokines activate immune defenses to eliminate infection, while anti-inflammatory cytokines limit tissue damage and promote healing (PubMed review). Pneumonia outcomes depend on how effectively these opposing forces are balanced over time. When this balance is maintained, inflammation supports infection clearance with minimal lung injury. When the balance is disrupted, inflammation itself becomes a major source of lung damage (National Heart, Lung, and Blood Institute).
Pro-inflammatory cytokines such as interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α) play a critical role in initiating immune defense during pneumonia. These cytokines increase blood vessel permeability, recruit immune cells to the lungs, and enhance the ability of immune cells to destroy pathogens (PubMed review). Data presented in the Data section (Table 1 and Table 2) show that individuals with more severe pneumonia exhibit markedly higher levels of IL-6 and TNF-α compared to those with milder disease. This pattern suggests that as pneumonia severity increases, the immune system shifts toward a stronger pro-inflammatory state. While this heightened response may improve pathogen clearance initially, sustained elevation of these cytokines increases the risk of excessive lung inflammation and tissue injury (PubMed review).
Anti-inflammatory cytokines such as interleukin-10 (IL-10) function to counterbalance this response by suppressing immune activity once the infection is controlled. IL-10 limits further recruitment of inflammatory immune cells and reduces the release of damaging substances in the lungs (PubMed review). Data comparing IL-6 and IL-10 levels across severity groups (Table 3) demonstrate that individuals with milder pneumonia tend to show a more proportional increase in anti-inflammatory cytokines relative to pro-inflammatory ones. This suggests that effective immune regulation, rather than immune strength alone, is associated with improved pneumonia outcomes. When IL-10 responses fail to rise adequately, inflammation may persist beyond what is necessary, increasing lung damage and delaying recovery (PubMed review).
The ratio between pro-inflammatory and anti-inflammatory cytokines provides important insight into pneumonia severity. Rather than examining individual cytokines in isolation, comparing their relative levels reveals how immune regulation shifts during disease progression. Data shown in the cytokine balance comparison tables (Table 4) indicate that severe pneumonia cases are characterized by a disproportionate increase in pro-inflammatory cytokines relative to anti-inflammatory cytokines. This imbalance supports the idea that lung injury in pneumonia often results from excessive immune activation rather than uncontrolled infection alone. A balanced cytokine response appears to allow inflammation to remain protective instead of destructive (PubMed review).
This pattern becomes even more pronounced when pneumonia occurs in individuals with prior viral respiratory infections. Data comparing moderate and severe COVID-19–associated pneumonia (Table 5) show a dramatic increase in IL-6 levels in severe cases, with a more modest change in TNF-α. This selective amplification of IL-6 suggests that previous viral exposure may alter how the immune system regulates specific cytokines (PubMed review). Elevated IL-6 has been linked to prolonged inflammation and increased lung tissue damage, indicating that prior viral infections may predispose individuals to exaggerated inflammatory responses during later pneumonia episodes (PubMed review).
Taken together, these data demonstrate that pneumonia severity aligns closely with the degree of cytokine imbalance rather than with infection presence alone. Mild pneumonia is associated with controlled increases in pro-inflammatory cytokines accompanied by timely anti-inflammatory regulation. Severe pneumonia is associated with sustained elevation of pro-inflammatory cytokines and insufficient suppression by anti-inflammatory signals. This imbalance leads to prolonged inflammation, alveolar damage, fluid accumulation, and impaired gas exchange. The tables presented in the Data section collectively support the interpretation that immune regulation determines whether inflammation remains beneficial or becomes harmful (National Heart, Lung, and Blood Institute; PubMed review).
The progression from moderate to severe pneumonia appears to reflect a gradual loss of cytokine control rather than an abrupt immune failure. As inflammation intensifies, cytokine signaling becomes increasingly skewed toward amplification rather than regulation. This explains why pneumonia outcomes vary widely among individuals and why similar pathogens can produce dramatically different clinical effects. Differences in cytokine balance help account for variability in lung injury, recovery time, and overall disease severity (PubMed review).
By examining cytokine levels across severity groups and infection histories, this research highlights the central role of immune balance in pneumonia outcomes. The data suggest that effective immune responses are defined not by the highest inflammatory activity, but by the ability to increase and decrease inflammation at appropriate times. This relationship between cytokine regulation and lung health forms a direct connection between the background research and the observed data trends, setting the stage for examining how prior viral infections further influence immune behavior and pneumonia severity (PubMed review).
Effects of Influenza and COVID-19 on Cytokine Regulation
Previous viral respiratory infections such as influenza and COVID-19 can significantly influence how the immune system regulates cytokines during later lung infections, including pneumonia. These viruses are known to trigger strong inflammatory responses in the respiratory system, activating immune cells and stimulating the release of pro-inflammatory cytokines. While this response is necessary to control viral replication, it can disrupt normal immune regulation in the lungs. After recovery, immune signaling pathways may remain altered, affecting how the body responds to future infections and increasing susceptibility to excessive inflammation (PubMed review; National Heart, Lung, and Blood Institute).
Influenza infection activates the immune system by increasing the production of pro-inflammatory cytokines such as IL-6 and TNF-α. This response helps eliminate infected cells but also places stress on lung tissue. Data presented in the Data section comparing individuals with prior viral infection to those without show that elevated pro-inflammatory cytokine levels are associated with increased pneumonia severity. These findings suggest that previous influenza exposure may prime the immune system to respond more aggressively during later pneumonia infections, increasing the likelihood of heightened inflammation rather than controlled immune activity (PubMed review).
COVID-19 has an especially strong effect on cytokine regulation and immune balance. During COVID-19 infection, the immune system may produce unusually high levels of inflammatory cytokines, particularly IL-6. This effect is reflected in the data comparing moderate and severe COVID-19 pneumonia cases (Table 5), where IL-6 levels increase dramatically as disease severity increases, while TNF-α levels rise more moderately. This pattern indicates that COVID-19 may amplify specific inflammatory pathways rather than causing a uniform immune response. Such selective amplification increases the risk of prolonged inflammation and lung tissue damage during subsequent pneumonia episodes (PubMed review).
The data also suggest that prior viral infections may interfere with the immune system’s ability to regulate inflammation effectively. Individuals with a history of influenza or COVID-19 show cytokine profiles that favor sustained pro-inflammatory signaling rather than balanced regulation. When pro-inflammatory cytokines remain elevated without sufficient counter-regulation, immune activity may persist beyond what is necessary to clear infection. This prolonged inflammatory state can damage the alveoli, increase fluid accumulation in the lungs, and slow recovery from pneumonia (PubMed review; National Heart, Lung, and Blood Institute).
Comparisons across severity groups reinforce the connection between viral history and pneumonia outcomes. Individuals without prior viral respiratory infections tend to exhibit more controlled increases in inflammatory cytokines during pneumonia, whereas those with prior influenza or COVID-19 show steeper increases in IL-6 and TNF-α as severity worsens. This trend supports the interpretation that immune history plays a critical role in shaping cytokine regulation and determining whether inflammation remains protective or becomes harmful (PubMed review).
Overall, the data indicate that influenza and COVID-19 can alter immune regulation in ways that persist beyond the initial infection. These alterations appear to increase the likelihood of cytokine imbalance during later pneumonia episodes, contributing to more severe inflammation and lung injury. By linking prior viral infections to changes in cytokine behavior, this section strengthens the connection between immune history, cytokine regulation, and pneumonia severity, setting the stage for an integrated understanding of how immune balance influences disease outcomes (PubMed review; National Heart, Lung, and Blood Institute).
Factors That Influence Cytokine Regulation
Cytokine regulation does not function the same way in every individual. Even when two people are infected with the same pneumonia-causing pathogen, their immune systems may respond very differently. These differences help explain why some individuals experience mild symptoms while others develop severe lung inflammation. Cytokine regulation is influenced by several interacting factors, including immune history, overall health, and how the immune system has responded to past infections. Together, these factors shape whether inflammation remains controlled or becomes harmful during pneumonia (PubMed review; National Heart, Lung, and Blood Institute).
One major factor influencing cytokine regulation is immune history. The immune system “remembers” past infections and adjusts future responses based on previous exposure. Individuals who have experienced respiratory viral infections such as influenza or COVID-19 may have immune cells that respond more quickly and more strongly when the lungs are infected again. While this rapid response can be helpful, it may also increase the risk of releasing too many pro-inflammatory cytokines too quickly. This can shift cytokine balance toward excessive inflammation, increasing the likelihood of lung tissue damage during pneumonia (PubMed review).
Overall health and immune system condition also play an important role in cytokine regulation. A healthy immune system is better able to increase inflammation when needed and reduce it once the infection begins to clear. When immune regulation is weakened or stressed, cytokine signaling may become less precise. This can lead to inflammation lasting longer than necessary or becoming more intense than required to control infection. Such disruptions increase the risk that inflammation will interfere with normal lung function rather than support recovery (PubMed review).
Age is another factor that can influence cytokine behavior. Younger immune systems are still developing, while older immune systems may respond more slowly or less accurately. These differences affect how cytokines are produced and regulated during infection. As a result, the timing and balance of inflammatory responses may vary between individuals, influencing how pneumonia progresses and how quickly healing occurs (PubMed review).
Environmental and biological differences also contribute to variation in cytokine regulation. Some individuals naturally produce higher levels of inflammatory cytokines, while others generate stronger anti-inflammatory responses. These differences can affect how well the immune system maintains balance during pneumonia. When cytokine signaling remains coordinated, inflammation helps clear infection without damaging lung tissue. When coordination is lost, inflammation may become excessive or poorly controlled (National Heart, Lung, and Blood Institute).
Taken together, these factors show that cytokine regulation is influenced by more than just the presence of infection. Immune history, health status, age, and individual immune responses all shape how cytokines behave during pneumonia. These influences help explain why pneumonia severity varies between individuals and reinforce the idea that immune balance, rather than immune strength alone, plays a critical role in determining disease outcomes. This understanding naturally leads into the broader interpretation of how cytokine regulation connects research findings to the observed data trends (PubMed review).
Connecting Cytokine Balance to Pneumonia Severity
Across this investigation, the immune response to pneumonia emerges as a system that depends heavily on balance rather than strength alone. From the initial activation of immune cells to the release of cytokines that guide inflammation, each stage of the immune process plays a role in determining whether pneumonia is resolved efficiently or progresses into a more severe condition. Rather than acting independently, immune cells, cytokines, and prior viral history interact in ways that shape lung inflammation and overall disease outcomes (PubMed review; National Heart, Lung, and Blood Institute).
The early sections of this project established how immune cells such as macrophages, neutrophils, and T cells respond to infection in the lungs by releasing signaling molecules that help coordinate defense. These signals, known as cytokines, were shown to be essential for recruiting immune cells and controlling pathogens. However, as later sections demonstrated, cytokines can become harmful when their levels rise too high or remain active for too long. This idea of regulation became a central theme linking immune response mechanics to real pneumonia outcomes (PubMed review).
When examining cytokines more closely, the distinction between pro-inflammatory and anti-inflammatory signals proved especially important. Pro-inflammatory cytokines such as IL-6 and TNF-α help the body fight infection, but the data presented in the tables show that elevated levels of these molecules are consistently associated with increased pneumonia severity. In contrast, anti-inflammatory cytokines act to reduce inflammation and protect lung tissue. The data section supports this balance-based interpretation, showing that severe cases are not defined by the presence of cytokines alone, but by how unevenly they are regulated (PubMed review).
This pattern becomes clearer when cytokine levels are compared across different severity groups. The tables demonstrate that patients with severe pneumonia exhibit sharply higher IL-6 levels than those with moderate illness, while TNF-α shows smaller but still meaningful increases. These differences suggest that excessive inflammatory signaling may contribute directly to lung damage, reduced oxygen exchange, and prolonged recovery. Rather than indicating a stronger immune system, the data imply that severe pneumonia reflects an immune response that has become poorly controlled (PubMed review).
The influence of prior viral infections, particularly influenza and COVID-19, further strengthens this interpretation. The section examining viral history highlights how past respiratory infections can alter cytokine regulation long after the virus itself has cleared. The tables comparing individuals with and without prior viral infection suggest that earlier exposure may prime the immune system to respond more aggressively, leading to higher cytokine release during later pneumonia infections. This connection helps explain why similar bacterial infections can produce very different outcomes in different individuals (PubMed review; National Heart, Lung, and Blood Institute).
Additional factors affecting cytokine regulation, including age, immune health, and environmental stressors, reinforce the idea that pneumonia severity cannot be attributed to a single cause. Instead, these factors influence how precisely the immune system turns inflammation on and off. When combined with the data trends observed across multiple tables, it becomes evident that pneumonia outcomes are shaped by a network of interacting variables rather than one dominant trigger (PubMed review).
Taken together, the research and data sections support a unified interpretation: pneumonia severity is closely linked to how well cytokine signaling is regulated during infection. A balanced immune response allows inflammation to clear pathogens while protecting lung tissue, whereas an imbalanced response increases the risk of severe disease. By connecting immune cell behavior, cytokine function, prior viral exposure, and measured data trends, this project highlights why immune balance, not immune intensity, is a key determinant of pneumonia outcomes (PubMed review; National Heart, Lung, and Blood Institute).
Data
Table 1
| Group | IL-6 | IL-10 |
|---|---|---|
| Non-severe Pneumonia (median at admission) | 19.98 | 3.68 |
| Severe Pneumonia (median at admission) | 44.07 | 1.99 |
Table 2
| IL-6 Mean (pg/mL) | TNF-α Mean (pg/mL) | |
|---|---|---|
| Healthy (Lima) | 16.2 | 25.9 |
| COVID-19 (Lima) | 48.3 | 61.6 |
| Healthy (Huaraz) | 67.3 | 89 |
| COVID-19 (Huaraz) | 97.9 | 120.6 |
Table 3
| IL-6 (pg/mL) | IL-10 (pg/mL) | TNF-α (pg/mL) | |
|---|---|---|---|
| COVID-19 without pneumonia | 1.85 | 5 | 6.35 |
| COVID-19 with pneumonia | 3.86 | 5 | 7.95 |
Table 4
| TNF-α (pg/mL) | |
|---|---|
| ARDS | 67 |
| Severe Pneumonia | 35 |
| Healthy Controls | 17 |
Table 5
| Severity Group | IL-6 (pg/mL) | TNF-α (pg/mL) |
|---|---|---|
| Moderate COVID-19 pneumonia | 2.9 | 7.5 |
| Severe COVID-19 pneumonia | 17.05 | 8.7 |
Table 6
| Group | Prior Influenza Infection | Average IL-6 Level (pg/mL) | Average TNF-α Level (pg/mL) |
|---|---|---|---|
| Group A | None | 25 | 18 |
| Group B | Influenza | 40 | 25 |
Final Summary Table
| Condition / Group | IL-6 (pg/mL) | IL-10 (pg/mL) | TNF-α (pg/mL) |
|---|---|---|---|
| Non-Severe Pneumonia | 19.98 | 3.68 | |
| Severe Pneumonia | 44.07 | 1.99 | |
| COVID-19 without Pneumonia | 1.85 | 5 | 6.35 |
| COVID-19 with Pneumonia | 3.86 | 5 | 7.95 |
| Moderate COVID-19 pneumonia | 2.9 | 7.5 | |
| Severe COVID-19 pneumonia | 17.05 | 8.7 | |
| ARDS | 67 | ||
| Controls (Healthy) | 17 |
Conclusion
This project shows that pneumonia outcomes depend more on immune balance than immune strength. Pro-inflammatory cytokines, such as IL-6 and TNF-α, are important because they help the body fight infection by activating immune cells and causing inflammation in the lungs. However, the research shows that when these cytokines stay high for too long or are not controlled, inflammation can damage the lungs instead of helping them heal. This damage affects the alveoli and makes it harder for oxygen to move into the bloodstream, leading to more severe pneumonia. Anti-inflammatory cytokines, such as IL-10, help control inflammation once the infection starts to clear. The data show that people with mild pneumonia usually have a better balance between pro-inflammatory and anti-inflammatory cytokines. This balance allows the immune system to remove the infection while protecting healthy lung tissue. In severe pneumonia cases, this balance is lost. Pro-inflammatory cytokines increase too much, while anti-inflammatory signals are not strong enough to reduce inflammation. The research also shows that having a past viral infection, such as influenza or COVID-19, can affect how the immune system responds during pneumonia. People with a history of these viruses tend to produce higher levels of pro-inflammatory cytokines, especially IL-6, during pneumonia. This suggests that previous viral infections may cause the immune system to react more strongly than needed, increasing lung inflammation and damage.
Overall, the results show that pneumonia becomes severe when cytokine balance is lost. A well-regulated immune response helps clear infection without harming the lungs, while an unbalanced response leads to lung damage and worse outcomes. This supports the idea that immune balance, influenced by cytokine regulation and past infections, plays a major role in determining pneumonia severity.
Citations
Please note that only the main citations that were used in the writeup are listed here. There were many other used, in the research itself.
“Pneumonia.” National Heart, Lung, and Blood Institute, U.S. Department of Health and Human Services, n.d., www.nhlbi.nih.gov/health/pneumonia. “Pathophysiology of Pulmonary Edema.” BCcampus Pressbooks, n.d., pressbooks.bccampus.ca/dcbiol2200/chapter/pathophysiology-of-pulmonary-edema/. StatPearls Publishing. “Pulmonary Edema.” StatPearls, National Center for Biotechnology Information, n.d., www.ncbi.nlm.nih.gov/books/NBK538282/. Ware, Lorraine B., and Michael A. Matthay. “The Acute Respiratory Distress Syndrome.” New England Journal of Medicine, vol. 342, no. 18, 2000, pp. 1334–1349. PubMed, pubmed.ncbi.nlm.nih.gov/9632909/.
Data Tables: https://pmc.ncbi.nlm.nih.gov/articles/PMC5131553/ https://link.springer.com/article/10.1186/s12879-022-07079-x https://pmc.ncbi.nlm.nih.gov/articles/PMC7389162/ https://pubmed.ncbi.nlm.nih.gov/10607801/ https://pmc.ncbi.nlm.nih.gov/articles/PMC7389162/ https://pubmed.ncbi.nlm.nih.gov/27905908/
Acknowledgement
I am sincerely grateful to my family for their constant encouragement throughout this project, especially my parents for motivating me to keep going, my sister for her support and feedback, my brother for his encouragement, and my dad for providing the materials I needed, as well as my grandpa, whose experience with pneumonia inspired this topic and gave my work personal meaning. I would like to thank my aunt for helping me understand complicated information written in some articles. I would also like to thank Ms. Bretner for giving me the opportunity to pursue this project, and Sarah Waqas for offering guidance and advice before I began, which helped me start this project with confidence and purpose.