The Effect of Various Antioxidants on the Rate of Oxidative Degradation of Levodopa in a Buffer Solution
Sriram Dokuparti
STEM Innovation Academy Jr. High SW
Grade 9
Presentation
No video provided
Hypothesis
Question: How can the inclusion of antioxidants produce an effect on the oxidation of levodopa in a chemical buffer solution?
Hypothesis:
Given levodopa's instability in an aqueous solution, I believe that the inclusion of antioxidants will produce an inhibiting effect of its rate of oxidation. Moreover, since antioxidants are cited with molecular systems that allow for the delocalization of electrons, any free radicals that may be produced would be stabilized by the antioxidant. However, I believe that this will not last for long because under the alkaline conditions, the antioxidant may also begin to lose power and levodopa would oxidize.Furthermore, I believe that all the antioxidants would produce a similar effect on the levodopa solution as they are cited with similar characteristics, although the mechanism of inhibition may slightly vary between each type:
Ascorbic Acid:
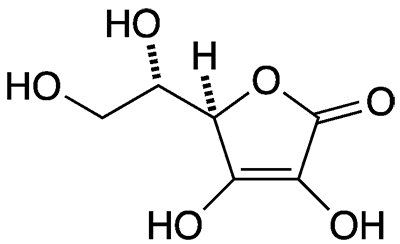 Analyzing the molecular structure of ascorbic acid, I believe that it will be effective in reducing the oxidation of levodopa due to the enediol group that can be found at the bottom of the aromatic ring. I believe that this structure (H-O-C=C-O-H) allows for excellent electron delocalization that enables the molecule to remain stable while donating electrons and protons. It may be the second most effective molecule in reducing the oxidation of levodopa when 2x more concentrated in molarity in comparison to the levodopa.
Analyzing the molecular structure of ascorbic acid, I believe that it will be effective in reducing the oxidation of levodopa due to the enediol group that can be found at the bottom of the aromatic ring. I believe that this structure (H-O-C=C-O-H) allows for excellent electron delocalization that enables the molecule to remain stable while donating electrons and protons. It may be the second most effective molecule in reducing the oxidation of levodopa when 2x more concentrated in molarity in comparison to the levodopa.
Citric Acid:
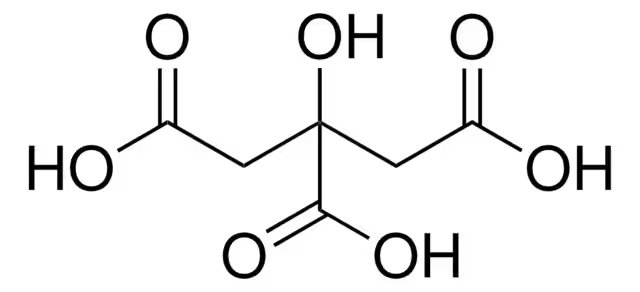 Analyzing the moelcular structure of citric acid, it can be hypothesized that this antioxidant will not be as efficeint as scorbic acid in slwoing the rate of oxidation as it does not contain a group that can remain extemely stable while donating electrons. I believe that this antioxidant however, will produce a potential effect by donating its protons to the levodopa molecule when it undergoes the oxidation mechanism in the presence of oxygen.
Analyzing the moelcular structure of citric acid, it can be hypothesized that this antioxidant will not be as efficeint as scorbic acid in slwoing the rate of oxidation as it does not contain a group that can remain extemely stable while donating electrons. I believe that this antioxidant however, will produce a potential effect by donating its protons to the levodopa molecule when it undergoes the oxidation mechanism in the presence of oxygen.
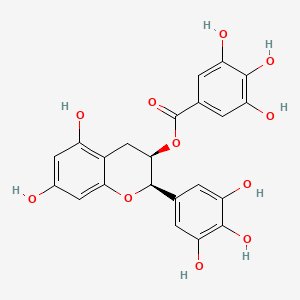 Analyzing the molecular structure, it can be inferred that this antioxidant would likely be the most most effective in reducing the rate of oxidation. As it contains multiple groups within its quad ring structure, it may donate a large amount of electrons and protons to any oxidized moleucles in the chemcal buffer to revert it back to its original state. Moreover, its gallate group may be of high effectiveness due to the crowding of atoms that it possesses, opening up the possibility for a major reducation of the oxidation rate.
Analyzing the molecular structure, it can be inferred that this antioxidant would likely be the most most effective in reducing the rate of oxidation. As it contains multiple groups within its quad ring structure, it may donate a large amount of electrons and protons to any oxidized moleucles in the chemcal buffer to revert it back to its original state. Moreover, its gallate group may be of high effectiveness due to the crowding of atoms that it possesses, opening up the possibility for a major reducation of the oxidation rate.
Research
Background Research (3 Sections):
Section A (Levodopa Chemistry and Biomedical Applications):
L-Dopa (levodopa), is noted as a precursor (a substance from which another is formed) to dopamine. Clinicians often use L-Dopa as a drug (replacement agent) for the Parkinson’s disease. It is mainly utilized to treat the bradykinetic symptoms that come with the Parkinson’s disease → “bradykinetic” refers to the slowed down movement, known as bradykinesia. This particular set of symptoms is prominent in neurological disorders, mainly Parkinson’s disease. One suffering from bradykinesia has trouble completing voluntary movements such as moving their arms and legs (voluntary movements of legs). Levodopa is typically prescribed to a patient that suffers from Parkinson’s and particularly to one that suffers fromParkinson’s of older age. Furthermore, L-Dopa maybe utilized for post encephalitic parkinsonism → a movement disorder with parkinsonian symptoms - slowness movements, muscle stiffness, and postural instability. Levodopa is typically prescribed to the individual that suffers from the Parkinson’s disease, typically when the disease becomes hard to control with anti parkinsonian drugs. The same drug is proved useful in the face of post-encephalitic parkinsonism (Parkinson’s disease that has developed because of severe brain infections or bacteria) and symptomatic parkinsonism (set of movement and brain abnormalities because of other pathologies rather than neurodegenerative causes). According to the National Library of Medicine, the administration of levodopa may either slowdown Parkinson’s disease or improve the Parkinson disease symptoms even after prescription is ceased. One common utilization of levodopa includes an offset or improving conditions for the symptom “restless-leg syndrome,” a prominent consequence of parkinsonism. Levodopa is subject to oxidative degradation due to its catechol functional group – an organic functional group that is characterized by a 1,2 dihydroxy benzene structure (class of organic compounds where two hydroxyl groups are attached to a benzene ring, which induces special properties of the substance). These properties include strong adhesions to surfaces, though not the most significant characteristic that sets levodopa apart from other catechol. These specific groups are susceptible to auto-chemical redox reactions, which involves the transfer of electrons between substances in an oxidation reaction. However, scientists can distinguish the rate of redox through the various attachments of chemicals to the benzene ring. Catechol may behave as antioxidants, scavenging free radicals, which is a crucial role in biological processes. When present in an aqueous solution or in the presence of oxygen, levodopa undergoes an automatic redox reaction to form an o-quinone (a group the two carbonyl groups)are attached to two adjacent carbon atoms, that are connected to a12-atom ring, such as the benzene ring. The concomitant generation of reactive oxygen species (highly reactive molecules containing oxygen which are naturally produced, sometimes in spite of a chemical reaction). For instance, reactive oxygen species are molecules that contain an unpaired electron, which consequently results in them reacting drastically with other substances in order to gain stability. Precursors to melanin such as those found in the redox of levodopa are particularly high in toxicity to neurons such as quinones. Oxygen concentrations severely affect the redox of levodopa, specifically higher concentrations accelerating the rate of redox.Studies note that oxygen tension is the core factor for levodopa oxidation, with few other factors accelerating it when paired with oxygen. The pH (potential of hydrogen) influences the oxidation speed of levodopa in a chemical buffer solution. More acidic solutions have proven to stabilize levodopa better due to the release of hydrogen ions in the redox reaction of levodopa. Conversely, the more alkaline the solution is, the acceleration of oxidation occurs.Studies confirm that when levodopa was dissolved in a solution with a pH of 10, levodopa lost approximately 20% of its content, within 1 hour, with solely 80% of the levodopa remaining.
Parkinson's Disease (Cause and Effects):
Parkinson’s disease is shown to be a chronic ailment (a disease orhealth effect that persists for three or more months, have the inabilityto be cured by traditional modern vaccines and injections) thatworsens over time within an individual which prominently affectstheir motor functions – leading to a defect in their ability to conductvoluntary movements. It is the second largest cause ofneurodegenerative disorders (the slow and continuous decay and deathof neurons, leading to a decline in brainfunction) after Alzheimer's disease. Many noticeable symptoms persistin Parkinson’s disease, such as muscle stiffness, and bradykinesia(slowed movement that may affect the individual’s voluntarymovements and automatic actions), along with postural instability withfurther exposure to the disease. According to the scientist’s diagnosisof recent studies, PD is predicted to increase to the senior and earlysenior population by over 30% in 2030. Parkinson’s disease is characterized by the loss of nigrostriataldopaminergic intervention (medical studies and researches thatattempt at developing strategies to monitor the function of thenigrostriatal pathway – a key dopaminergic (anything related todopamine) pathway that controls voluntary movement by projectingfrom the substantia nigra pars compacta (region in the mid-brain containing dopaminergic neurons, which are crucial for motorcontrol and are deeply impacted by the Parkinson’s disease) to thedorsal striatum (a major component in the brain responsible forcontrolling learning, movement, and cognition). However, theneurodegeneration of Parkinson’s disease does not solely impact thecells located within the nigral dopaminergic neurons, but also othercells part of neural connections in the brain. Thus, since theParkinson’s disease affects such as large population of neurons, asingle diagnosis of the disease is impossible to the complexity of thebrain and the scale of effect Parkinson’s causes. Dopamine neuronal deaths are shown to the root of Parkinson’sdisease’s effect, leading to a-synuclein aggregates, mitochondrialdysfunction, reactive oxygen species (ROS), apoptosis, andneuroinflammation. A-synuclein is a presynaptic neuronal protein (acomplex group of molecules in the pre-synaptic neuron that are crucialfor the transmission of signals to other neurons, serving purposes thatinclude organizing synaptic structures, docking or triggering therelease of neurotransmitters, and neuronal development) and isdetailed to misfold during the period of time when the individual hasthe Parkinson’s disease, often forming Lewy Bodies (abnormalclumps of protein that build up inside the neuron) and is thereforeassociated with neuroinflammation (an inflammatory response in thenervous tissue of the brain and the spinal cord, triggered by factorssuch as disease, and neurological infections). Scientists note that suchresponse from the neurons within the brain is the prime factor thatallows scientists to gain a basic knowledge whether the patient suffersfrom the Parkinson’s disease. The formation of Lewy Bodies of a-synuclein proteins in pre-synaptic neuron terminals primarily impairsthe neurotransmitter release of dopamine in dopaminergic neurons,leading to synaptic dysfunction and the eventual degeneration ofdendritic spines (small protrusions – structure that extends beyond thesurface – from neurons that receive most excitatory input). This occursbefore the cell body dies itself, leading to dysfunction. Manyindividuals believe that like other cells, neurons may undergo mitosis,where the cell divides to result in two identical cells with the samenumber of chromosomes as the parent cell. However, neurons arespecialized cells that may not undergo mitosis, lasting for the lifespanthat the human lives. Thus, the early death of neurons may bedetrimental to the patient. Dopamine is a neurotransmitter produced in the brain and plays a rolein the body’s reward system and bodily functions, including memory,movement, motivation, moods etc. High or low dopamine levels withinthe brain are linked to Parkinson’s disease, ADHD, and restless legsyndrome. As deduced prior, dopamine is a monoamineneurotransmitter used in the brain, again, for the functions of specifictypes of signals. However, dopamine also acts as a hormone. Dopamine,norepinephrine, and epinephrine maintain a similar chemicalstructure. These hormones are made by the adrenal glands and is alsoa neurotransmitter released by the hypothalamus (small butessential component of brain, responsible for releasing hormones thatregulate certain bodily processes. Functioning as a hormone, dopamine is released in the bloodstream.Although epinephrine & norepinephrine are the main hormonesresponsible for the “flight” or “fight” response, dopamine does play asmall role within it.
Antioxidants (Basics):
Ascorbic Acid: Essential water-soluble nutrient, also known asVitamin C and must be consumed through food as the body cannotnaturally synthesize it. It plays a key role in the production ofcollagen, a vital protein that is required for healthy skin, tendons,ligaments, etc. Citric Extract: Acts as an antioxidant to prevent cells fromoxidative stress, and its benefits include enhancing nutrientabsorption in the body, supporting the immune system, andpromoting liver and skin health. EGCG: Abundant antioxidant derived from green tea that is knownfor its antioxidant and anti-inflammatory properties, and studied forpotential health benefits such as cancer chemoprevention,improving cardiovascular health, and supporting weightmanagement, particularly through lowering LDL (low-densitylipoprotein).
Variables
Manipulated Variable:
The type of antioxidant utilized in the redox reaction when dissolved in the chemical buffer at a pH of 10. The various antioxidants include ascorbic acid, citric extract (antioxidant), and EGCG (found in green tea. Make up 50-80% of the catechins).
Responding Variable:
The pH acidification of levodopa in the solution containing antioxidants. As the redox forms multiple products, the observation of such provides insight in the product – dopachrome, which is a precursor to melanin. The pictures will be taken across a 16-hour experimental period at an appropriate timestamp.
Controlled Variables:
| Controlled Variable | Reason for Constancy | Method of Constancy |
|---|---|---|
| Oxygen Intensity | The rate of redox reactions with unstable substances, particularly oxidizing ones have an acceleration of reaction with higher oxygen intensities. Thus, the reaction would inevitably occur faster and introduce a new manipulated variable: How does oxygen intensity play a role in the redox reaction. Moreover, the oxygen intensity may also affect the antioxidant and achieve unexpected or unreliable results. | The approximate oxygen intensity will be monitored as the experiment will take place on a singular day, without minor fluctuations in oxygen intensities across different days. All the trials will be performed on the same day and administered at relatively similar times to minimize the uncertainty of varying oxygen concentrations. |
| pH (Power of Hydrogen) | The redox reaction for levodopa releases hydrogen ions that acidify the solution and accelerate the oxidation, When compared with pH of 2, the oxidation of levodopa decreased to almost 0% even after the 50 hour mark. However, a pH pass 9-10 requires lab materials and environments due to the corrosiveness of the substances. | A specific amount sodium bicarbonate and carbonate will be dissolved in a selective amount of chemical buffer solution to decrease the pH to make it more alkaline to an approximate pH of 10 to ensure an accelerated redox reaction for the levodopa. |
| Temperature | Higher temperatures were studied to produce a faster redox reaction on levodopa due to the faster kinetic energies of the particles between the reactants, which ultimately led to a faster reaction. Thus, differentiation between the two tests would again, introduce a new manipulated variable into the experiment: temperature. | All the tests will be performed in a setting with room temperature conditions throughout the sixteen hour experimental condition. |
Procedure
Part A: Preparation (Area):
- Preferably, attempt to achieve a room with mitigated air circulation from external sources for maximum accuracy as it prevents the uneven escaping or entering of oxygen within the room which may interfere unevenly with the levodopa oxidation process.
-
Ensure a shelf or organization utensil is within reach during all times of the experiment to place equipment in an organized manner to prevent any misplacements or poor organization.
-
Ensure any moisture is removed off of the utensil using a dry and slightly rough paper towel to ensure a dry workspace.
-
Attempt to achieve a table, preferably one larger than 39” x 24”.
-
Place a table cloth over the table. A laminated table cloth is recommended for ease of cleaning any stains or fluids that may have spilled.
-
Ensure any moisture is removed off of the table using a dry and slightly rough paper towel to ensure a dry workspace.
-
Ensure a clear and organized notebook is present to record observations during the experiment
- If the area of completion is present with a centralized heating system, ensuring the thermostat is set to 20 o Celsius.
Part B: Preparation (Chemical & Quantifiable Equipment):
- Place all the materials that are utilized in the process of the experiment on the shelf or organization utensil and label each with a sticky note (preferably use a pen to ensure clear visibility)
- Ensure conditions above are constant: temperature is centralized to 20 degrees Celsius and ventilation of sunlight is present, preferably shining indirectly on the table to ensure light conditions are present for later steps.
Part C: Preparation (PPE - Personal Protective Equipment):
- Utilizing lab-safety gloves retrieved, ensure they are moisture free.
- Tie the lab apron retrieved around the front area of the body and ensure protection is secured. Additionally utilize plastic eye goggles to prevent the entering of any irritants into the eye that may cause complications.
- Additionally utilize a full-sleeve t-shirt and mask to prevent the inhalation of fine particles into the body, as well as any irritants from contacting the skin.
Part D: Preparation of Buffer Solution (Section 1):
- Ubox the pH meter with the three separate pH calibration sachets (4.00, 6.86, and 9.18). Place these aside away from the main workspace for the time being
- Unbox the six pieces of borosilicate glass beakers, each with a capacity of 250 mL and increments up to that bracket.
- Utilizing a bottle of distilled water, selectively pour 250 mL into each borosilicate glass beaker and wait for 15 seconds to allow for it to settle. Repeat for the second glass beaker.
- Pour the 6.86 pH sachet into one beaker and stir clockwise or counterclockwise until the contents dissolve. Utilize a sticky note and label this beaker.
- Repeat with the other beaker by adding the 9.18 pH sachet to effectively bracket the desired pH of 10 for the carbonate buffer that will be prepared at a later step.
-
Over the chemical waste disposal (stainless steel container), rinse pH meter probe gently with distilled water and lightly dab with paper towel.
-
Ensure that when dabbing with a paper towel, light pressure is applied and the towel is folded to ensure that it is soft and cushioned. Additionally, ensure that the towel that may be rough is not moved across the meter’s glass.
-
Place the probe of the pH meter within the solution containing a pH of 6.86. Press “CAL” for 5 seconds until the screen on the meter shows the 6.86 pH reading.
- Repeat the rinsing process and place probe in the 9.18 pH solution
- Quick press “CAL” once after pH meter shows 9.18
- Repeat the rinsing process and set pH meter aside for time being. Ensure the probe is well-protected and isolated from any materials that may damage its ability to read measurements.
Part D: Preparation of Chemical Buffer Solution (Part 2):
- Unbox the kitchen scale and place it near the central workspace, along with the sodium carbonate and bicarbonate.
- In a stainless steel bowl that is moisture-free and clean, add 800 mL of distilled water, measured using the 100 mL graduated cylinder.
- Allow for the distilled water to settle for approximately 10-15 seconds.
- Using the tray provided with the kitchen scale, precisely add 0.62 g of sodium carbonate and 1.19 g of sodium carbonate into the 800 mL of distilled water and stir in a cc or ccw direction until all solids are dissolved.
- Using the calibrated pH meter, test the pH of the solution precisely. It should be at about 9.95-9.96.
- Following this, add 200 mL of distilled water to top up the solution to 1L of sodium carbonate buffer at 0.02 moles.
- Store this in a dark environment, preferably the organization utensil.
- Utilizing the graduated cylinder, split the 1 L of sodium carbonate buffer solution into the remaining four beakers that have a capacity of containing 250 mL of solution.
- Using the sticky notes, label each beaker differently, with the following order of labels: “Controlled - No antioxidant,” “Ascorbic Acid - 8mM,” “Citric Acid - 8mM,” “EGCG - 8mM.”
Part E: Preparation of Usable Chemicals and Antioxidants (Part 1):
- Unbox the ascorbic acid and citric acid which are readily in powdered form.
- Weight 0.21 g of ascorbic acid and set aside in a paper towel, with a sticky note labelled “Ascorbic Acid Pre-Configured Measurement”
- Weigh 0.23 g of citric acid and repeat the steps such that are similar for ascorbic acid.
- For EGCG, crush the capsules with a mortar and pestle into a fine powder in which the grains are <1 mm in length. Ensure that the total weighed powder totals to 1.1 g, effectively allowing for 8 mM.
- Set aside in a paper towel and repeat steps such that are similar to ascorbic acid.
Part E: Preparation of Usable Chemicals and Antioxidants (Part 2):
- Dissolve each selective antioxidant in three of the remaining four 250 mL beakers and stir in a direction continuously for 20 min, two at a time.
- Quickly unbox the mucuna prurien capsules and crush into fine powder using a rinsed mortar and pestle to ensure that the overall weight on the kitchen scale is 0.78 g.
- Utilize a rinsed kitchen scale tray to weigh \~0.2 g of mucuna prurien powder and dissolve first into the controlled (no antioxidant) beaker.
- Stir for 20 min in a continuous direction and utilize a sieve to strain any remaining particles and pour the purified solution containing infused levodopa into three separate 50 mL capacity beakers.
- Repeat for all tests and ensure that there is a 30 sec difference between the mixing and straining.
Part F: Testing Phase:
- For the first three cycles, attempt to take pictures and take pH meter readings after continuous rinse every 30 min.
- Proceed by taking readings every hour for the next three cycles
- Proceed by taking readings every two hours for the rest of the cycles with continuous rinses of the pH meter.
- Take observations and set up for analysis in the future steps.
Observations
Quantitative Observations:
Pre Data: Color Index:
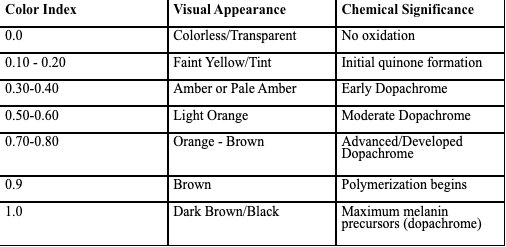
Original pH (Buffer Solution):
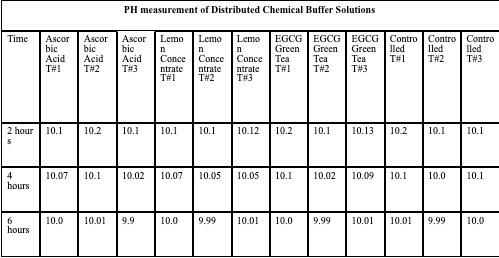
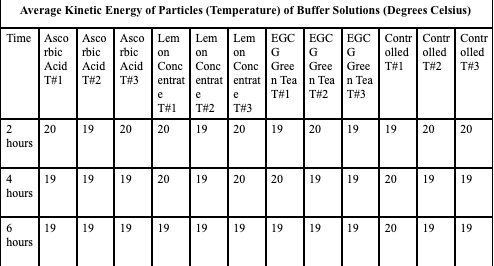
Temperature of Buffer Solution:
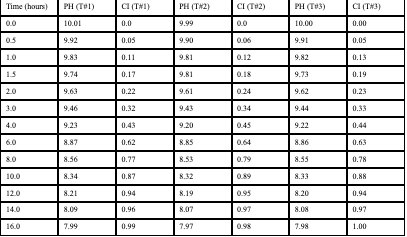
Raw Data of Levodopa Oxidation (Control Group – No antioxidants
Raw Data (Ascorbic Acid Group Solution)
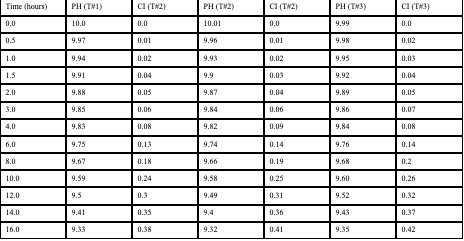
Raw Data (Citric Acid Group Solution)
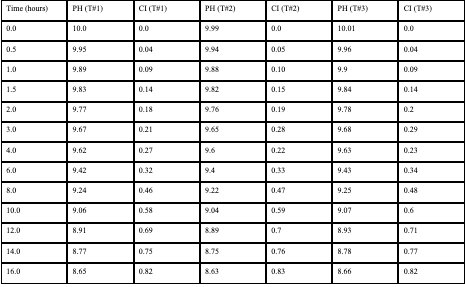
Raw Data (EGCG Group)
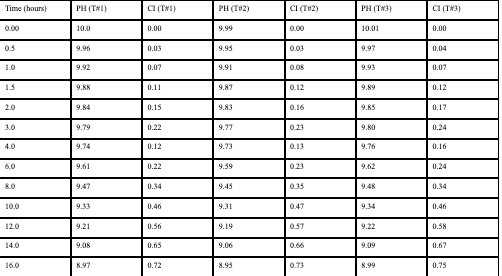
Raw Data (Test Averages):
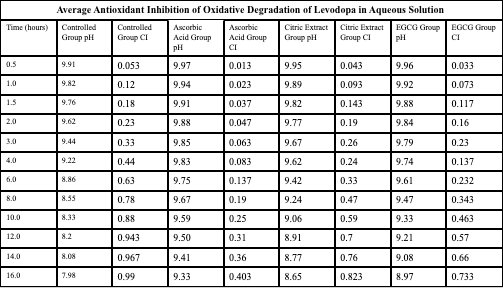
Qualitative Observations:

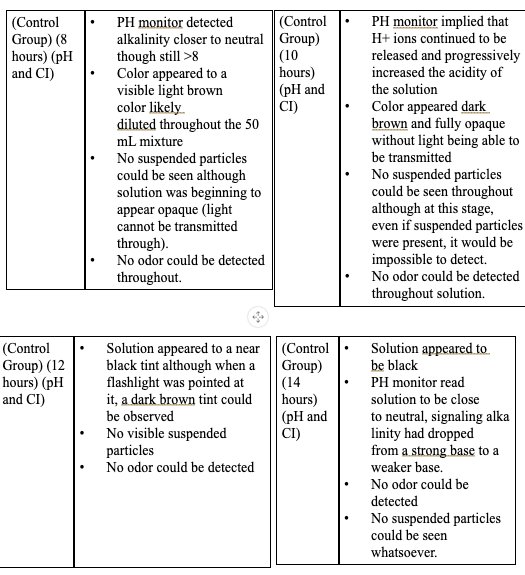
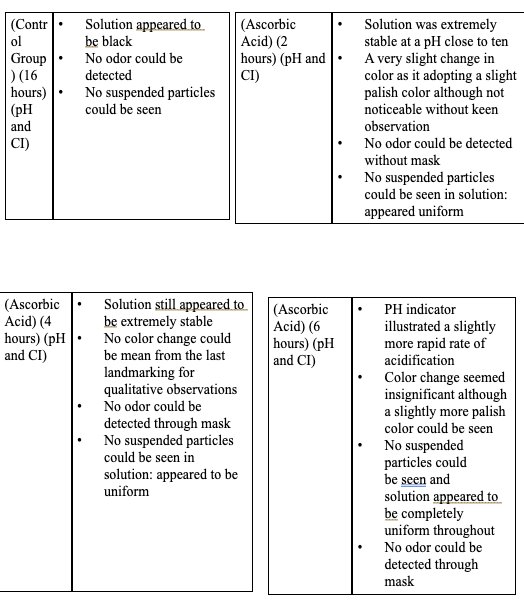
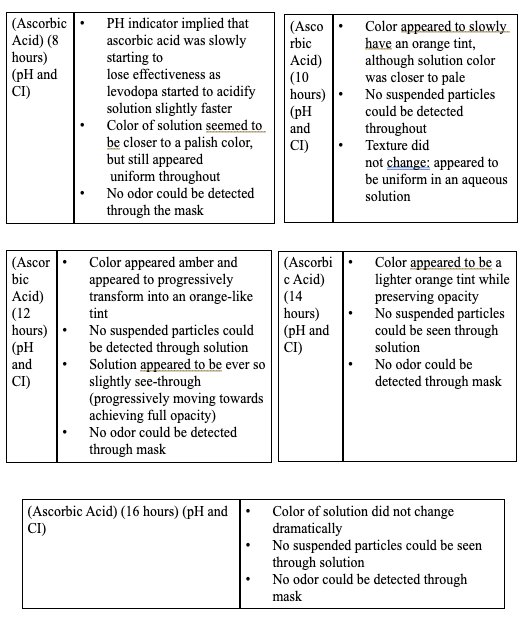
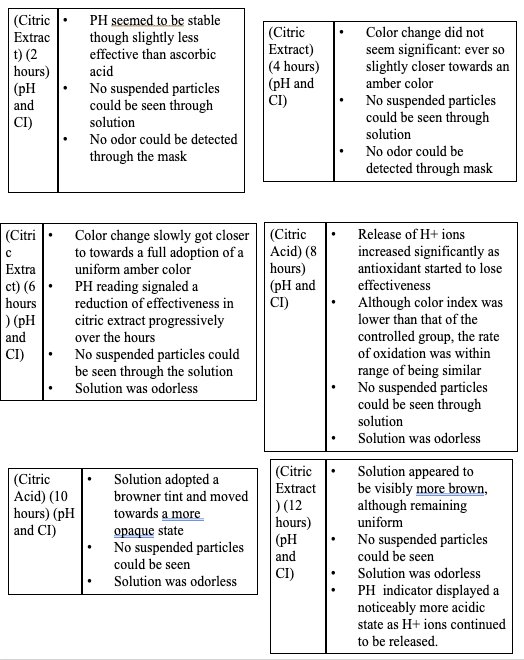
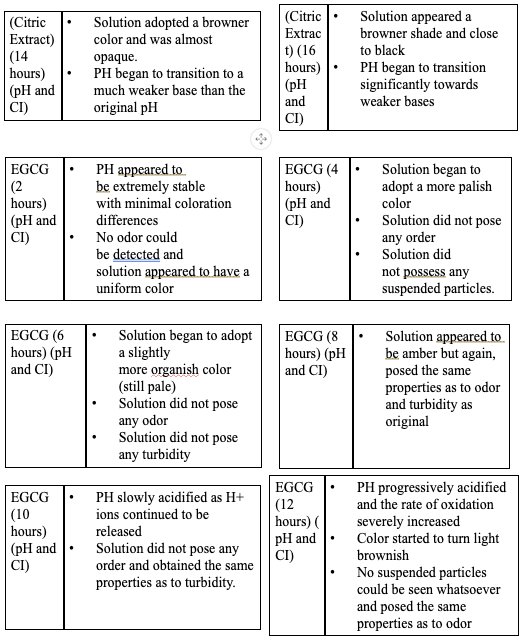
 |
|---|
Analysis
Analysis Section 1: Understanding Results:
Section 1a: Core Mechanism (Control Group):
Levodopa undergoes severe auto-oxidation triggered by a redox reaction inthe presence of oxygen. While being accelerated by an increase in alkalinity,the specific mechanisms can be understood to prove how the color index ofthe solution increases with an opposite reaction of the pH decreasing. At apH of 10, the hydroxide ions in the alkaline solution take protons from thephenolic –OH group in the solution to develop a catecholate (deprotonatedphenolic –OH group (dianion)). The two deprotonated phenolicoxygen groups form double covalent bonds with carbon atoms that aremembers of the phenyl ring. Ultimately, this causes the groups tobecome electrophilic as within the double bond, the oxygen atoms pulls theelectronegativity towards itself – oxygen has more protons than carbon in amore compact atom size allowing for a more effective nuclear charge –leaving carbon partially positively charged and oxygen electron-rich. Meanwhile, the amine group (functional group with a lone pair) in alevodopa molecule is described as NH2. Amine groups are electron-rich,meaning that upon forming a covalent bond with an atom, the group hasextra electrons. This group remains unprotonated in the presence of analkaline pH, it remains unchanged with an extra lone pair – nucleophilic.It is this first reaction of oxidation that forms dopaquinone, providing apalish tint to the solution. Dopaquinone has two carbonyls. However, only of these carbonylgroups is positioned close enough for intramolecular attack. Here, thegeometric effect takes place where the amine group can only reach thecarbonyl group that it is closest to. At a high pH, the amine isnucleophilic. The lone pair on the amine attacks the electrophilic carbonylcarbon that is closest to it. This forms the C-N covalent bond. Because thereacting atoms are part of the same molecule, it is known as anintramolecular nucleophilic attack. This attack naturally forms a five-membered ring that is the most stable ring in thesystem. This ring closure product is a molecule knownas leucodopachrome. Leucodopachrome is also very palish in color. After leucodopachrome is formed, the environment still contains oxygen which is astrong oxidizing agent because it is naturally a strong electron receiver. The C-N bond implies that the nitrogen officially becomes part of a ring, where thehydrogen atom is replaced by a carbon atom – nitrogen can only maintain threecovalent bonds at a time. When the lone pair attacks the electrophilic group, itbecomes part of a ring which results in it having a covalent bond to two carbonatoms and one hydrogen atom. Thus, the NH2 becomes NH. It looses the hydrogenatom. Leucodopachrome has two hydrogen atoms that may still be removed as well aselectrons that may donated to the oxygen. Oxygen atomsoxidize leucodopachrome to accept the electrons that are preventing the moleculefrom being fully conjugated – sigma bonds do not allow for the delocalization ofelectrons as it tightly holds electrons between two atoms. The hydrogen protons arethen released into the solution where the carbon atoms that had to form 4 sigmabonds can rehybridize to form the lowest energy arrangement possible. Originally,carbon maintained four sigma bonds that utilized 1s and 3p orbital in the valenceshell to develop 4 sigma bonds. However, when the sigma bond between hydrogenand carbon is removed, one p orbital is unhybridized. Two adjacent carbon atomseach donate one p orbital to result in a pi bond being form. This is the transition fromsp3 to sp2. This leaves 3 sigma bonds and 1 unhybridized p orbital which overlapswith the neighboring p orbitals or its adjacent carbon atom. This side-side overlap isknown as pi bonding. As electrons always choose the lowest energy arrangementpossible, a delocalized pi bonding system has a far lower energy arrangement thansigma bonds. The electrons therefore move into the overlapping p orbitals and formpi bonds. As this spreads out to the entire ring, the resulting molecule is known asdopachrome. According to scientific principles, delocalized electrons can absorbvisible light which results in the orange/brownish pigment. A dopachrome molecule still possess an phenyl –OH group. In an alkaline solution, thismeans that the group is deprotonated (OH- accepts protons) making the electrons stayas lone pairs to the oxygen atom. Subsequently, the lone pair on the oxygen atomoverlaps with the carbon atom’s p orbital, resulting in the electrons being delocalizedaround the whole ring. Consequently, conjugation is extended and light absorptionincreases. Ultimately, it results in a darker pigment being revealed: browner. However, with increased conjugation, electron-shifts may occur that may lead to a pibond being formed at the carbon that forms a single bond between the ring and anadjacent carbon outside the ring. For this to occur, electron-density is pulled away fromthe carbon that bonds with the carboxyl group, weakening it. Eventually, this breaks offas carbon dioxide. However, the remaining electrons in the bond re-arrange to form thesecond double bond in carbon dioxide. Simultaneously, the electrons in the ring shift tocreate a pi bond. This tautomerization is what allows for a more conjugated moleculethat can absorb visible light to a higher degree. Thus, it contains brownish/blackeningpigments. The resulting molecule is known as DHI (dihydroxy indole). From here, the carbon atoms that are adjacent to the nitrogen (C2 or C3) lose anelectron by oxidation. It happens specifically here as carbons that lose electronshere can delocalize better. Thus, a carbon-centered radical is formed. The unpairedelectron can be delocalized across the 5-membered ring, into the lone pair of nitrogen,or the conjugated pi system in the phenyl ring. This delocalization stabilizes the radical.According to scientific principles, a stabilized radical is far more likely to undergopolymerization as the system can never be fully conjugated and paired (missing oneelectron). Subsequently, a process known as radical coupling occurs where two DHIstabilized radicals meet and the unpaired electrons form a covalent bond. In thiscircumstance, the carbon that formed the center radical pairs with the carbon fromanother molecule. There is uncertainty to which carbon atom forms the radical, but thepossibilities are C2-C2, C2-C3, or C3-C3. As this continues, more center radicals areformed on other DHI molecules and form covalent bonds. The DHI molecules thenform a covalent bond with its adjacent molecule, allowing the indole structures tooverlap, allowing for an increased electron delocalization. It is this because of thisdelocalization that melanin can absorb a wide-range of light (UV and visible) toprovide a blackish color.
Section 1b: Ascorbic Acid:
Doing a quick micro view, the redox reaction for levodopa may only occur if themolecule loses electrons and the oxygen in the solution gains electrons. This is knownas the redox pair. If levodopa cannot donate electrons, the redox cannot be initiated. Anascorbic acid molecule is built in a method that when in mobile form, ithas conjugated system that loses electrons easily and stabilizes radicals. Putting intoperspective, the ascorbic acid is sacrificing its electrons to stabilize free radicals. Ascorbic acid has an enediol system meaning it contains a structure represented by thefollowing: HO-C=C-OH. Unlike the stronger acid levodopa, this structure allowsascorbic acid to be highly delocalized because the double bond between carbons allowsfor electron density to be shared through resonance. When the enediolgroup looses electrons, the unpaired electron can spread over the double bond, bothhydroxyl groups, and the entire ring. This forms the ascorbic radical, which is stabilizedby resonance among the entire ring and conjugation between the double bond andoxygen atoms. This stabilized radical is what allows for easy electrondonation whereas levodopa cannot form a radical that is this stable because of itscomplex carbon bonding's that prevent the atom from reaching sp2. Moreover, ascorbic acid reduces the oxidation of levodopa the strongest due to itsbehavior if levodopa oxidizes to form a quinone. Since quinones areelectrophilic, ascorbic acid donates an electron to the quinone to make it a semi-quinoneradical. The gaining of this electron makes the oxygen on the quinone strongly basic.Thus, it immediately grabs a proton from the surrounding water or from the hydroxylgroup on the enediol system. Following this, ascorbate donates a second electron toconvert the quinone into a catechol oxygen. The second quinone group now becomessemiquinone and is extremely basic. Ascorbate thus donates another proton to the groupand reverts it back into a catechol. However, because this process is not continuouslysustainable, the levodopa does eventually oxidize into its redox products asthe ascorbic acid continues to sacrifice itself. Once the enediol system in ascorbic acid is depleted, it is known as dehydroascorbicacid. Since it is fully oxidized now, levodopa is the main electron donor in the solution.Thus, it undergoes natural oxidation. However, this oxidation is not as fast as a solutionwithout ascorbic acid because when oxygen is present at the start, it consumes oxygen,temporarily lowers the oxygen concentration, and delays the buildup of quinones. Thus,when levodopa starts to oxidize, it is oxidizing in a system where there is less dissolvedoxygen concentration than in a fresh solution.
Section 1c: Citric Acid:
The fundamental method in which citric extract inhibits the oxidation of levodopais that it donates protons to the solution around it. A citric acidmolecule contains three carboxylic groups (-COOH) which are each capable ofdonating one proton. In an alkaline solution, the hydroxide ions readily acceptthese protons. When these protons bond with the hydroxide ions, the solutiondevelops H2O as it moves towards becoming a neutral solution. Citricextract contains an abundance of citric acid molecules which each contribute toneutralizing the pH. Revisiting the reaction between levodopa and OH-, a limit onthe abundance of hydroxide ions in the water reduce the rate at which this ion canreact with the catechol and produce a catecholate. However, in this experiment, avery small amount of citric acid was applied to the solution, implying that thedeprotonation of the levodopa molecule is very slightly delayed.
Section 1d: EGCG:
EGCG has multiple different phenolic rings in that when placed in the same solution aslevodopa, the gallate ring (three –OH groups) naturally is oriented to be parallel tolevodopa’s catechol. When these two rings align, polarization of the electronclouds occur – the EGCG cloud pushes and pulls on levodopa’s electron cloud, causinga shift in electron density. As EGCG’s gallate ring is full of hydroxyl groups, the ring ismore conjugated and electron-rich than levodopa’s catechol ring. Thus, the two cloudsrepel each where EGCG’s electron density is directed in the opposite direction oflevodopa’s. Consequently, levodopa’s catechol ring becomes slightly poor. As thestability of a ring is dependent on how well it can delocalize the electrons, when theelectron density is pulled away from the ring, the oxygen atom in the hydroxyl group oflevodopa becomes unstable. Thus, it holds onto the proton more tightly as forming theO- ion is expensive. Forming the ion is more unlikely as the oxygen atom itself is lessacidic, matching the pH of the solution more closely. However, over time, the –OH bonds in the gallate group start to become deprotonatedby the surrounding environment, resulting in more electrophilic rings. Over time,EGCG cannot shield levodopa and repel the electron density away fromthe catechol’s ring. Moreover, over time, it cannot help prevent deprotonation andlevodopa eventually oxidizes. However, this is far more effective than citric extract butslightly less effective than ascorbic acid as it does not continuously repair quinones andrevert them back into catechol's.
Analysis Section 2: Trend Analysis:
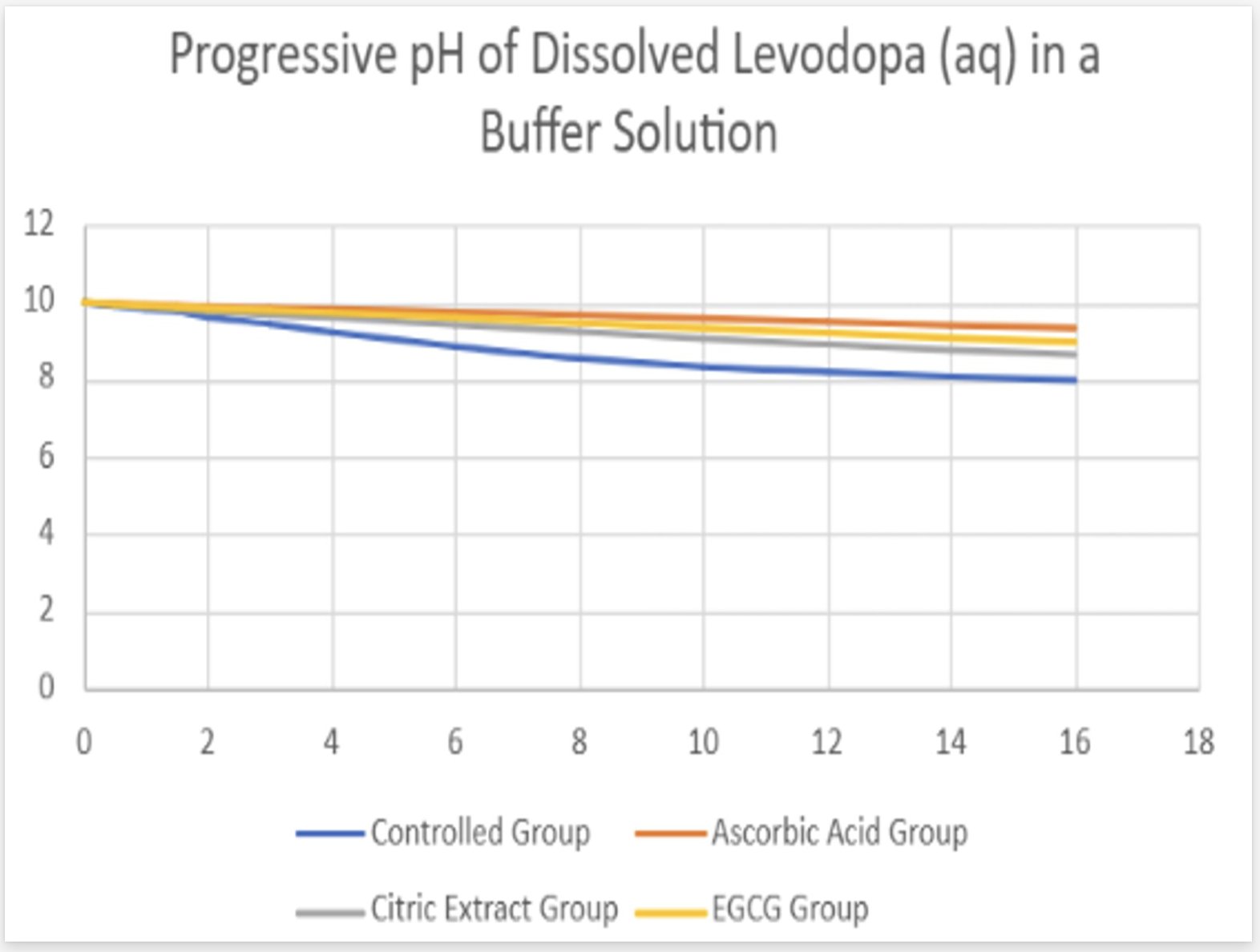
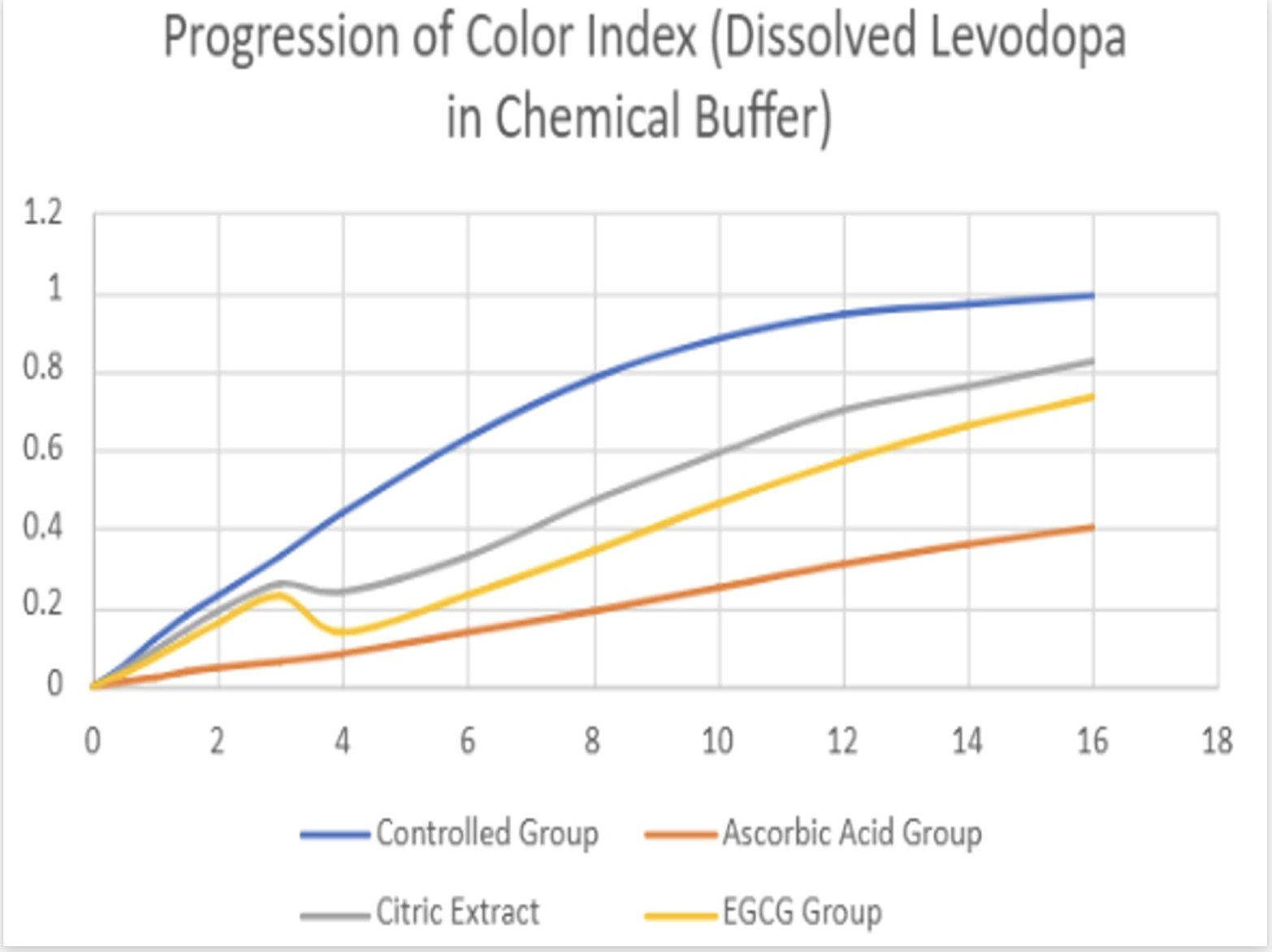
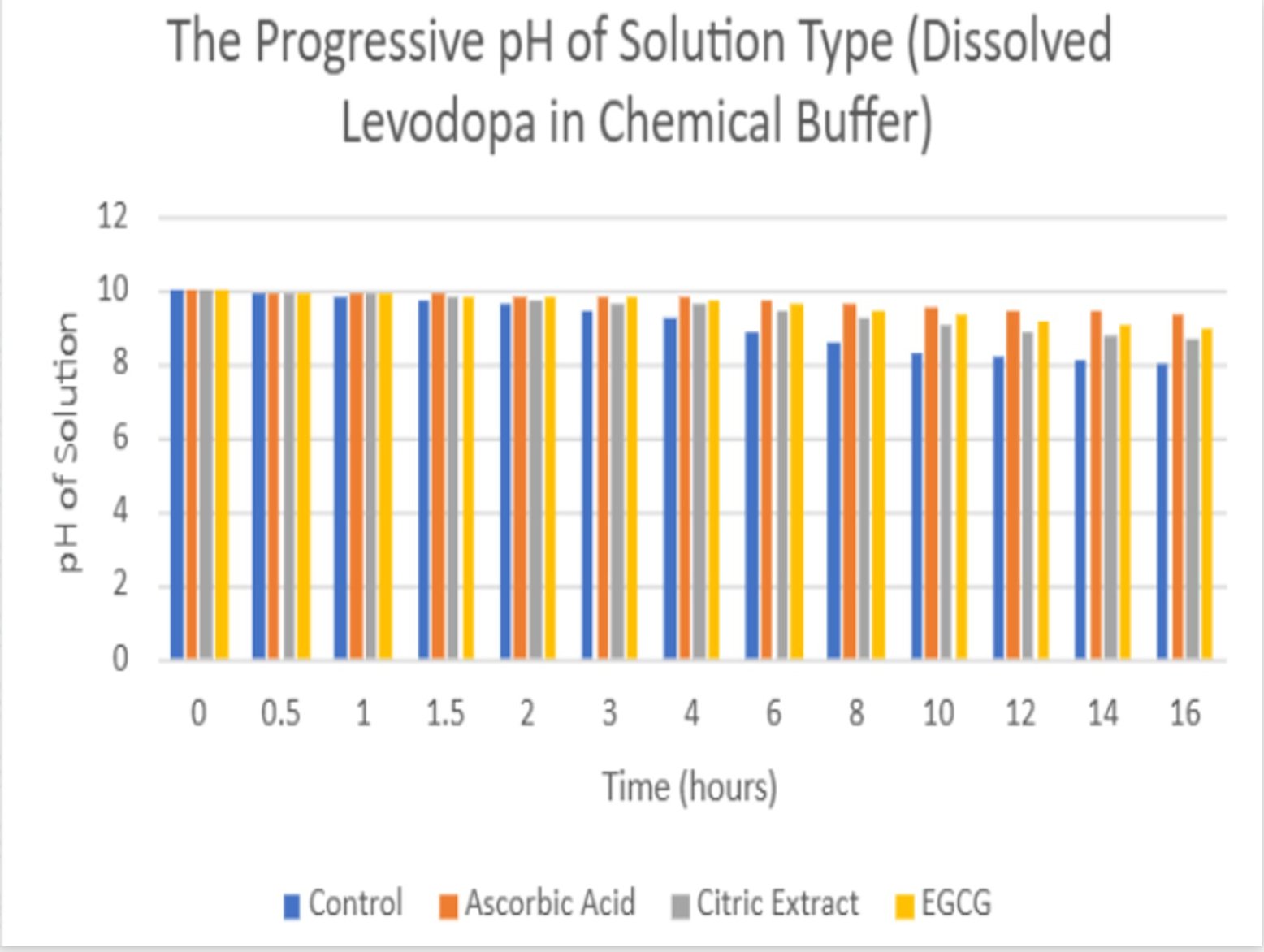
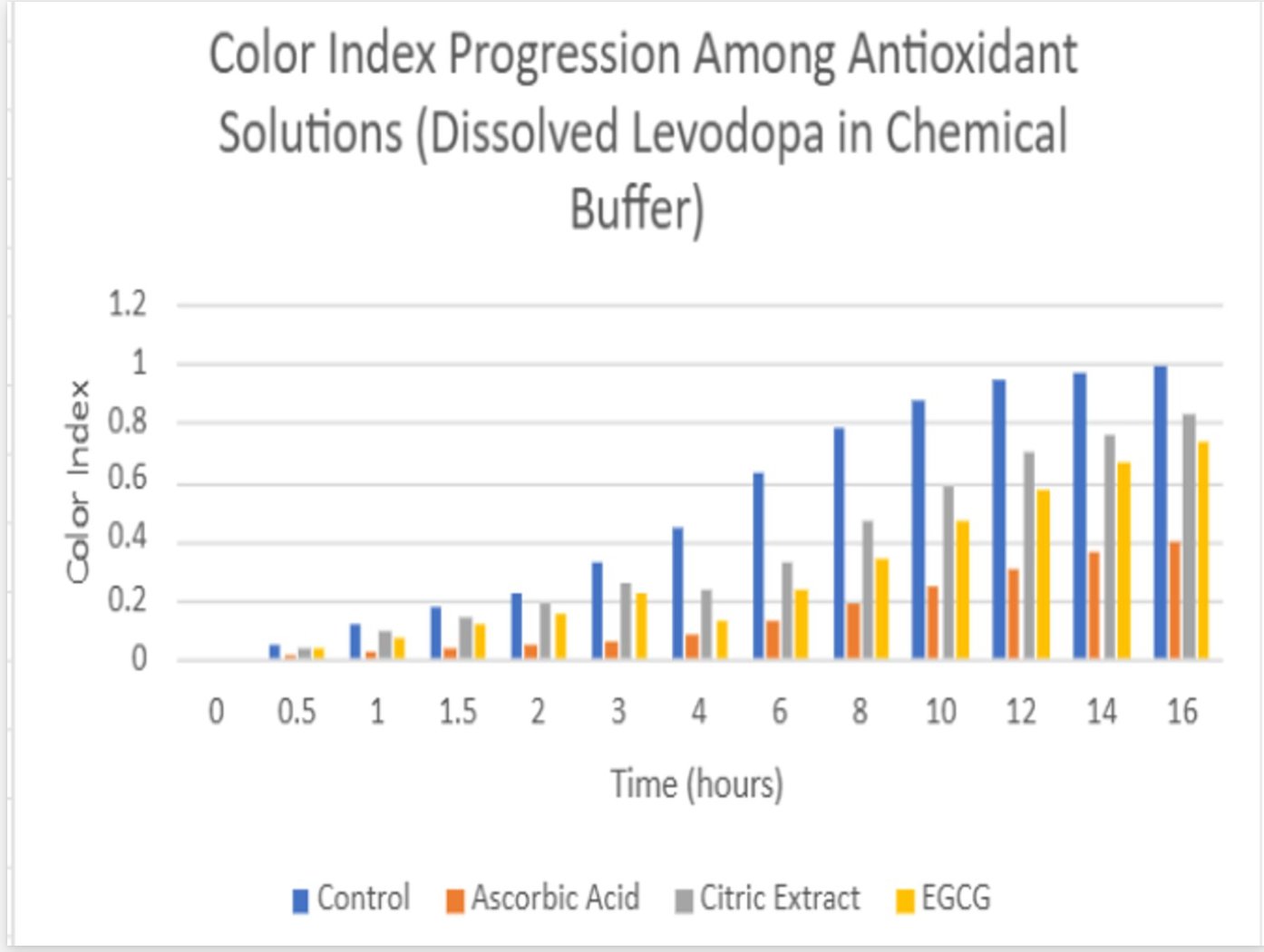
- The solution containing levodopa without any antioxidants proved to havethe fastest oxidative degradation because of deprotonation, catecholateformation, and the subsequent redox reactions.
- Based on the data, all the antioxidants proved to have a reducing effect onthe oxidative degradation of levodopa due to their own respectiveprocesses, with ascorbic acid being the most effective, EGCG being thesecond most effective, and citric extract being the least effectiveantioxidant.
- Moreover, the graphs illustrate that the greatest inhibition on the browningeffect after 16 hours is ascorbic acid due to its effectiveness in preventingthe early quinone formation of levodopa.
- Over all of the trials, it can be concluded that ascorbic acid had thestrongest inhibition effect due to its sacrificial abilities, whereas EGCGhad a similar early strength but later reduced in effectiveness due to itsown subsequent deprotonation.
- Citric extract, though posing the ability to donate a vast number of protonsto rapidly neutralize the solution, was not added in enough of a significantconcentration to prove effectiveness in inhibiting theoxidative degradation as effectively.
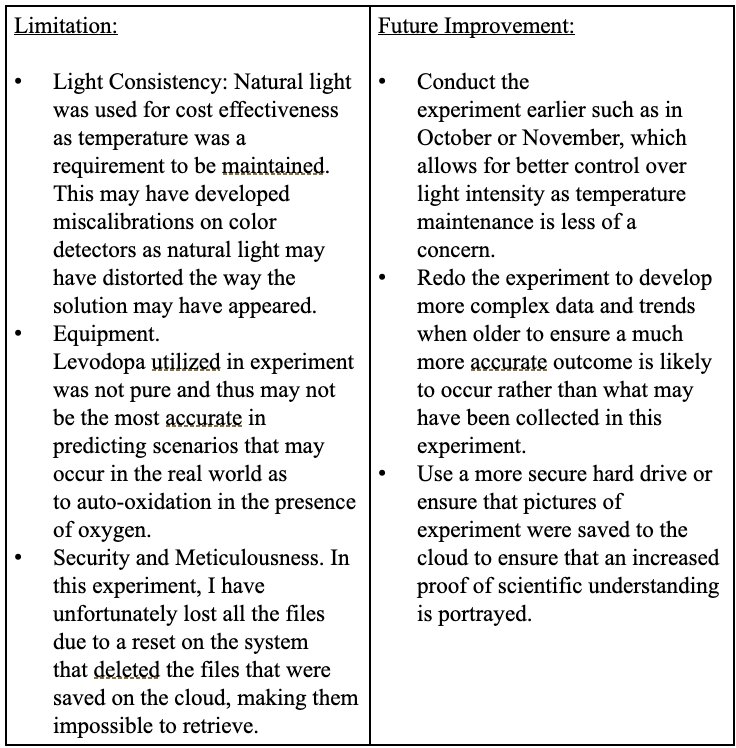
Section 3: Experimental Limitations:
Conclusion
Antioxidants play a large role in inhibiting the oxidative degradation oflevodopa. Especially in a more alkaline condition, antioxidants delay thequinone production of levodopa, leading to its subsequent product andeventually, melanin-like precursors. The purpose of this experiment was toverify and further prove scientists’ observations on how antioxidants candelay the oxidation of levodopa to ensure that the patient of the levodopatreatment may receive their treatment without harmful quinones. Provingthis once more, the data can be referenced to understand that the strongestantioxidant of the three – ascorbic acid, demonstrated an average59% inhibition of oxidative degradation in compared to the controlled group.Revisiting Section 1 of the analysis again, we can understand this was likelya result of the sacrificial behaviors of the ascorbic acid, readily reverting anyquinones of levodopa back into a catechol by providing the necessaryprotons and electrons from its enediol system. This project can be partiallysuccessful as although the data was collected, there could have been bettercontrol over the temperature and light exposure towards the tests and trials,which may have opened better accuracy.
Application
Levodopa is utilized in parametrical settings and control Parkinson’s disease in thepatient by serving as a dopamine supplement. However, it is chemically unstablebecause it auto-oxidizes in the presence of oxygen and forms quinones and melanin-like products. However, the data collected illustrates that antioxidants are effective ininhibiting the oxidation of levodopa in aqueous solutions. Potentially, this can beapplied to medical settings where antioxidants can help preserve shelf life byreducing oxidation during storage, preserve potency, and the catechol form.Previously, we already discussed how ascorbic acid possesses the ability to revert anyquinones back into the catechol forms of levodopa. Thinking broadly, this canminimize uncertainty into oxygen leaks in medical settings to allow the treatment tobe as successful in controlling the various symptoms of Parkinson’s disease suchas bradykinesia and muscle stiffness. In some medical settings, levodopa is administered to the patient through injectiontubes and infusions. It is in these settings that the drug solutions may accidentally beexposed to oxygen over time during extended treatments. The data collected suggeststhat these issues may potentially be minimized, or provide added benefits asantioxidants to the rest of the body. Moreover, other molecules such as epinephrine are chemically susceptible tooxidation and thus require proper storage and transportation. This data may suggestthat antioxidants may further be applied to those molecules to additionally increasethe purity that it is provided to patients at. To push this project further, the specific amount of antioxidant concentration could beresearched and experimented with in an aqueous solution to understand how theconcentration of antioxidant would impact the levodopa concentration in a neutralsetting. It could be tested whether it would overpower the levodopa or impact itschemical structure in a way that it is defective.
Sources Of Error
Sources of Error:
| Limitation | Improvement: |
|---|---|
|
|
Citations
- Kalia, Lorraine V., and Anthony E. Lang. “Parkinson’s Disease.” The Lancet, vol. 386, no. 9996, 2015, pp. 896–912.
- Poewe, Werner, et al. “Parkinson Disease.” Nature Reviews Disease Primers, vol. 3, 2017, article 17013.
- Dauer, William, and Serge Przedborski. “Parkinson’s Disease: Mechanisms and Models.” Neuron, vol. 39, no. 6, 2003, pp. 889–909.
- Obeso, José A., et al. “Missing Pieces in the Parkinson’s Disease Puzzle.” Nature Medicine, vol. 16, 2010, pp. 653–661.
- Surmeier, D. James, et al. “Dopamine in the Basal Ganglia.” Neuron, vol. 76, no. 1, 2012, pp. 90–103.
- National Institute of Neurological Disorders and Stroke. “Parkinson’s Disease Information Page.” NIH, 2023.
- Nolte, John. The Human Brain: An Introduction to Its Functional Anatomy. 7th ed., Elsevier, 2016.
- Standring, Susan, editor. Gray’s Anatomy: The Anatomical Basis of Clinical Practice. 42nd ed., Elsevier, 2020.
- Purves, Dale, et al. Neuroscience. 6th ed., Oxford UP, 2018.
- Kandel, Eric R., et al. Principles of Neural Science. 6th ed., McGraw-Hill, 2021.
- Blumenfeld, Hal. Neuroanatomy through Clinical Cases. 3rd ed., Sinauer Associates, 2021.
- Nieuwenhuys, Rudolf, et al. The Human Central Nervous System. Springer, 2008.
- Hille, Bertil. Ion Channels of Excitable Membranes. 3rd ed., Sinauer Associates, 2001.
- Bean, Bruce P. “The Action Potential in Mammalian Central Neurons.” Nature Reviews Neuroscience, vol. 8, 2007, pp. 451–465.
- Südhof, Thomas C. “Neurotransmitter Release.” Cold Spring Harbor Perspectives in Biology, vol. 5, no. 1, 2013.
- Katz, Bernard. The Release of Neural Transmitter Substances. Liverpool UP, 1969.
- Kandel, Eric R. “The Molecular Biology of Memory Storage.” Science, vol. 294, 2001, pp. 1030–1038.
- Meiser, Johannes, et al. “The Biochemistry of Neurotransmitters.” Cell Communication and Signaling, vol. 11, 2013.
- Beaulieu, Jean-Martin, and Raul R. Gainetdinov. “The Physiology, Signaling, and Pharmacology of Dopamine Receptors.” Pharmacological Reviews, vol. 63, no. 1, 2011, pp. 182–217.
- Missale, Claudia, et al. “Dopamine Receptors.” Physiological Reviews, vol. 78, no. 1, 1998, pp. 189–225.
- Rang, H. P., et al. Rang & Dale’s Pharmacology. 9th ed., Elsevier, 2019.
- Cotzias, George C., et al. “Modification of Parkinsonism—Chronic Treatment with L-Dopa.” New England Journal of Medicine, vol. 280, 1969, pp. 337–345.
- Fahn, Stanley. “The History of Levodopa as Parkinson’s Disease Therapy.” Movement Disorders, vol. 23, 2008.
- Olanow, C. Warren, et al. “Continuous Dopaminergic Stimulation in Parkinson’s Disease.” The Lancet Neurology, vol. 5, 2006, pp. 677–687.
- Graham, Daniel G. “Oxidative Pathways for Catecholamines in the Presence of Metal Ions.” Molecular Pharmacology, vol. 14, 1978.
- Graham, Daniel G., et al. “The Autoxidation of Dopamine.” Journal of Biological Chemistry, vol. 253, 1978.
- Ito, Shosuke. “The Reaction of Catechol with Quinones.” Pigment Cell Research, vol. 16, 2003.
- Napolitano, Alessandra, et al. “Oxidation Chemistry of Catecholamines.” Chemical Research in Toxicology, vol. 24, 2011.
- Segura-Aguilar, Juan, et al. “Dopamine Oxidation and Neurotoxicity.” Journal of Neurochemistry, vol. 129, 2014.
- Bisaglia, Marco, et al. “Dopamine Quinones.” Neurochemistry International, vol. 62, 2013.
- Halliwell, Barry, and John M. C. Gutteridge. Free Radicals in Biology and Medicine. 5th ed., Oxford UP, 2015.
- Valko, Marian, et al. “Free Radicals and Antioxidants.” The International Journal of Biochemistry & Cell Biology, vol. 39, 2007.
- Finkel, Toren, and Nikki J. Holbrook. “Oxidants, Oxidative Stress and the Biology of Ageing.” Nature, vol. 408, 2000.
- Rice-Evans, Catherine, et al. “Antioxidant Properties of Phenolic Compounds.” Trends in Plant Science, vol. 2, 1997.
- Packer, Lester, and Enrique Cadenas. Handbook of Antioxidants. 2nd ed., CRC Press, 2001.
- Carr, Anitra C., and Balz Frei. “Toward a New Recommended Dietary Allowance for Vitamin C.” The American Journal of Clinical Nutrition, vol. 69, 1999.
- Buettner, Garry R. “The Peeling of the Onion: Ascorbate and Prooxidant Chemistry.” Free Radical Biology and Medicine, vol. 25, 1998.
- Du, Jun, et al. “Mechanisms of Ascorbate-Induced Cytotoxicity.” PNAS, vol. 109, 2012.
- Mandel, Silvia, et al. “Green Tea Catechins and Neuroprotection.” Journal of Neurochemistry, vol. 88, 2004.
- Weinreb, Orly, et al. “Neuroprotective Molecular Mechanisms of EGCG.” Neurotoxicity Research, vol. 18, 2010.
- Sang, Shengmin, et al. “Stability of Tea Polyphenols.” Journal of Agricultural and Food Chemistry, vol. 53, 2005.
- Winterbourn, Christine C. “Toxicity of Iron and Hydrogen Peroxide.” Toxicology Letters, vol. 82, 1995.
- Kehrer, James P. “The Haber–Weiss Reaction.” Free Radical Biology and Medicine, vol. 25, 2000.
- Valko, Marian, et al. “Metals, Toxicity and Oxidative Stress.” Current Medicinal Chemistry, vol. 12, 2005.
- Snyder, Lloyd R., et al. Introduction to Modern Liquid Chromatography. 3rd ed., Wiley, 2010.
- Harris, Daniel C. Quantitative Chemical Analysis. 9th ed., W. H. Freeman, 2016.
- Skoog, Douglas A., et al. Principles of Instrumental Analysis. 7th ed., Cengage, 2018.
- Motulsky, Harvey, and Arthur Christopoulos. Fitting Models to Biological Data Using Linear and Nonlinear Regression. Oxford UP, 2004.
- Ito, Shosuke, and Kenji Wakamatsu. “Chemistry of Melanins.” Pigment Cell Research, vol. 16, 2003.
- Prota, Giuseppe. Melanins and Melanogenesis. Academic Press, 1992.
- Gandhi, Kavita R., and Abdolreza Saadabadi. “Levodopa (L-Dopa).” StatPearls [Internet], StatPearls Publishing, 17 Apr. 2023, NCBI Bookshelf, www.ncbi.nlm.nih.gov/books/NBK482140/. Accessed 12 Feb. 2026
- “How to Calibrate a pH Meter for Accurate Results.” ScienceInsights, https://scienceinsights.org/how-to-calibrate-a-ph-meter-for-accurate-results/? Accessed 12 Feb. 2026
Credits:
- Parents and Sibling: Assisted in supporting me mentally into gaining the motivation required to successfully complete the project.
- Science Fair Coordinator(s): Assisted in pointing my project towards the right direction and setting up the school fair for me to participate in.
- Microsoft Copilot: Assisted in breaking down extremely complex concepts of organic chemistry to ensure that I could slowly understand mechanisms effectively and accurately. I later searched through some primary sources that covered these concepts to ensure that inaccuracy of understanding was mitigated.
Acknowledgement
I extend my heartful gratitude towards many individuals that assisted me in the completion of this project. This involves my parents and sibling who acted as mental support to allow me to gain motivation to participate in the fair and gain insightful knowledge into the applications of levodopa and how we currently use it. Moreover, the science fair coordinators at my school were extremely helpful in directing my project towards the right direction to ensure that I did not take detours that prevented me from analyzing or collecting data. Adding on, I acknowledge microsoft copilot as a useful tool in assisting in the completion of this project, primarily through the assistance with understanding complex ideas of the organic chemistry. To ensure that the information curated was accurate, I searched through primary sources that covered these concepts that allowed me to ensure information was accurate while finsihing the project effectively and efficiently.

