Which household liquid causes metal to corrode the fastest?
Aanya Bhandari
Annie Gale School
Grade 9
Presentation
No video provided
Hypothesis
Hypothesis: I think that the products I'm going to use will corrode because, for corrosion to happen, my nail that I’m going to use will lose electrons. But I also think that it’s not going to work because the nail I will use will probably not be accurate to the household tools that I am comparing to.
Research
What does corrode mean? Corrode is a scientific term used to describe destroying or damaging metals through a chemical action. Corrode has 2 meanings:
- Destroy or damage through chemical action
- Destroy or weaken something moderately.
What are the main causes of corrosion? The main causes of corrosion are: Electrochemical (also can be defined as redox): oxidation: Metal atoms LOSE electrons, causes to form ions
Reduction: other substances or oxygen GAINS those electrons and forms oxide (similarly to rust).
Environmental factors: Moisture and humidity: water typically acts like an electrolyte which speeds up the oxidation process.
Salts and chloride: Salt water and marine air significantly increase the corrosion rates.
How can you prevent corrosion? As we know, corrosion is awfully dangerous. Here are some ways to prevent corrosion:
Keep smaller objects indoors: If you have a bike or a car or any small item that can easily corrode, put them inside. Corrosion is caused by air and moisture so it's highly recommended to put small objects inside.
Environmental actions: When metal comes in contact with water or salt water, it slowly starts to degrade. If you have items that aren’t corrosion resistant, it is a good idea to shelter them to prevent corrosion.
Choosing corrosion resistant metal Choosing a corrosion resistant metal such as aluminum or stainless steel can briefly impact your corrosion chances.
How can corrosion impact household equipment?
Corrosion can have a huge impact on household equipment causing failure regarding functions, deterioration, and health hazards.
Failures in tools that have been corroded can easily prevent fixtures. For example, if your dishwasher has been corroded (internal) can lead to significant damage such as leaks.
Deterioration (mainly revolving around aesthetics) can leave and show an unpleasant stain and other sights such as flaking, and overall show a very unpleasant appearance in the house or property.
Health hazards contribute to corrosion because, if you were to drink water, unaware that your pipe is slowly corroding, you can potentially drink water with a terrible metallic taste and if it gets bad, it can be a serious health hazard.
What are the benefits of corrosion prevention?
The benefits of corrosion prevention are: Reduced maintenance costs: preventing corrosion can prevent high costs repairment that would be needed for highly valued tools.
Increased efficiency: when an item is not corroded, it can be more efficient.
Increased reliability: without corrosion occurring, fixing malfunctioning objects would be way more easier than a tool that is corroded and hard to work with.
Variables
Responding variable: looking for which nail will corrode the fastest Manipulated variable: the household liquid Controlled variable: same glass, same screwdriving nails, same temperature.
Procedure
Procedure: First get 2 clear glass cups. Secondly, put 2 aluminum screwdriving nails in the cup. Third, pour table salt water inside the cup with the nail inside. Once you put the amount your satisfied with, put the same amount of pure white vinegar in the other glass cup with the nail still in the cup. Finally, wait for 3 days to notice a change.
Observations
Observations:
Day 1:
This one is vinegar this one is salt water
So far, I haven't noticed any corrosion.
Day 2
Table salt water vinegar
I noticed that the vinegar had completely corroded the nail but the table salt water didn't even affect the nail at all.
Day 3 (last day)
I finished my experiment and found out that the nail in vinegar had completely corroded and I realized that the table salt water didn't even corrode.
Analysis
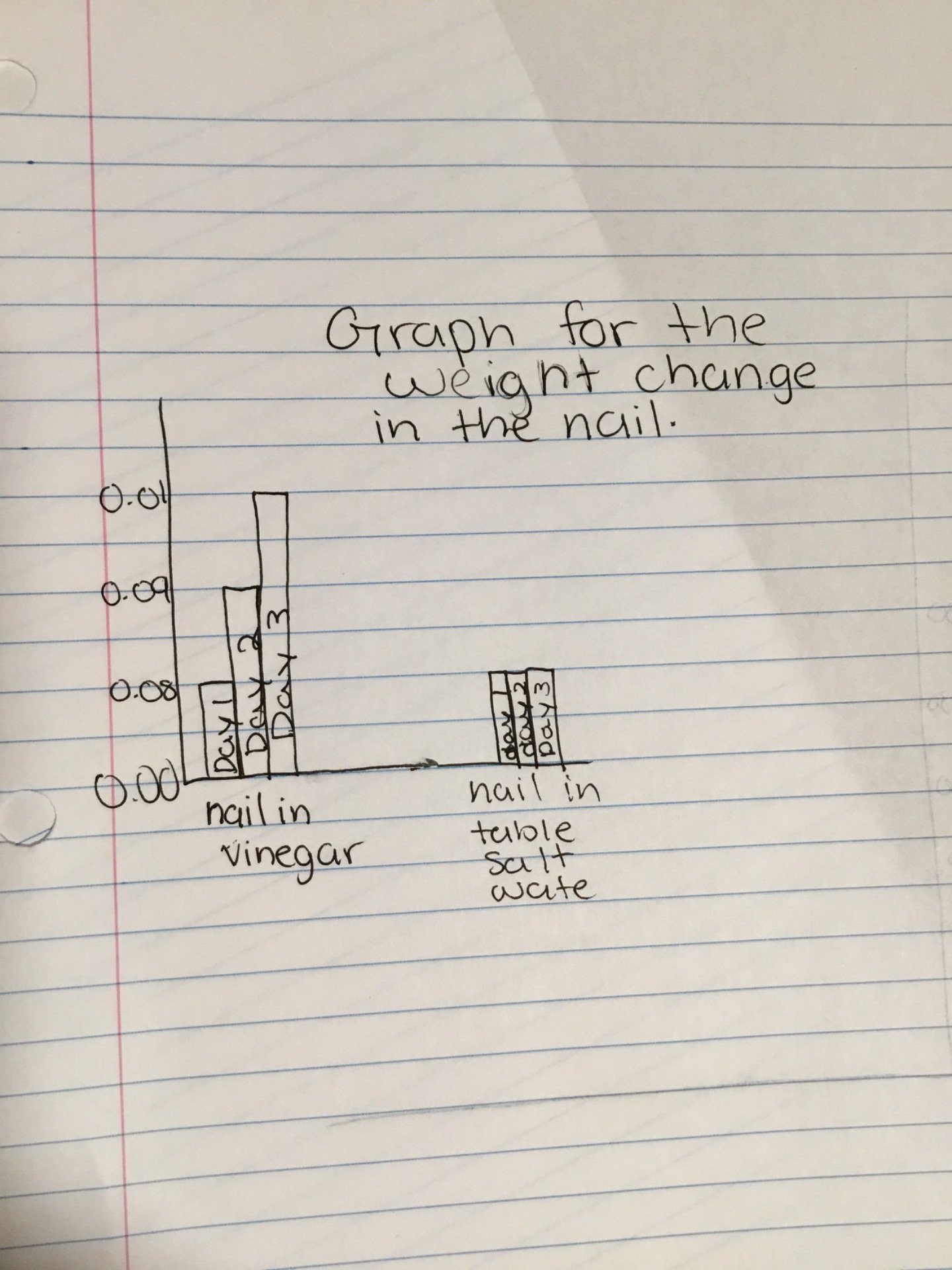 Analysis:
From what I observed, I realized that the nail in the vinegar increased its weight. Both of the nails started at 0.08kgs but as the days progressed, the nail in the vinegar became 0.09 kgs on day 2 and then 0.01kgs on day 3.
I created my graph on paper.
Analysis:
From what I observed, I realized that the nail in the vinegar increased its weight. Both of the nails started at 0.08kgs but as the days progressed, the nail in the vinegar became 0.09 kgs on day 2 and then 0.01kgs on day 3.
I created my graph on paper.
Conclusion
Conclusion: Overall, I would say that my hypothesis was somewhat correct. I say this because, in my hypothesis, I stated that it will happen because it will lose electrons. But I also stated that it may not happen because the material in the nail was “not like household utensils.” My hypothesis also stated that corrosion will occur, and it actually did.
Application
Since corrosion is a huge thing that can make our tools or items less efficient, understanding what types of materials that can corrode by liquids or air can help us stabilize your materials. If there were to be a case where someone doesn’t know how to handle corrosion, with the knowledge you have, you can help them.
Sources Of Error
My biggest source of error was that, I should have used actual good materials that could have demonstrated my experiment more better. I also think that I should have made a more better hypothesis and should have made the experiment more critical considering of the grade I am.
Citations
https://www.youtube.com/watch?v=Y0s44Wcrwak&t=113s
What is Corrosion?
How To Prevent Corrosion
The Best Practices to Prevent Rust and Corrosion in Your ...
Top 5 Benefits of Corrosion Resistant Materials - SLPSB
Acknowledgement
I would like to deeply acknowledge the people that supported me throughout this project. I would like to thank my parents, my teacher, my friends, and everyone who gave me faith into this fair.

