The Salty Solution
Oscar Pilecki
STEM Innovation Academy Jr. High SW
Grade 7
Presentation
No video provided
Hypothesis
Increasing salt concentration within the electrolyte will reduce internal resistance and increase voltage under load due to higher ionic conductivity, but excessive salt may increase corrosion and reduce overall efficiency.
Research
Definitions:
Magnesium-Air Fuel Cell
In a magnesium-air fuel cell with a salt (NaCl) electrolyte, the primary electrochemical reaction is the anodic oxidation of magnesium and the cathodic reduction of oxygen to produce electricity and magnesium hydroxide precipitate. This reaction is also accompanied by parasitic corrosion that produces hydrogen gas.
- Electrochemical: Relating to or involving both electricity and chemistry.
- Electrolyte: A neutral or saline solution (e.g. Salty water) that allows ions (not electrons) to travel between the anode and cathode.
- Oxidation: A chemical reaction resulting in the loss of electrons at the anode.
- Reduction: A chemical reaction resulting in the gain of electrons at the cathode.
- Anode: Negative electrode by which the electrons leave a device (Fuel)
- Cathode: Positive electrode by which the electrons enter a device (Oxygen)
- Parasitic Corrosion: An unwanted chemical reaction.
- Precipitate: Precipitate is an insoluble solid that emerges from a liquid solution. It can make a clear solution look opaque.
Fuel Cell Types – Quick Comparison:
Acronym Definitions:
PEMFC – Proton Exchange Membrane Fuel Cell SOFC – Solid Oxide Fuel Cell AFC – Alkaline Fuel Cell PAFC – Phosphoric Acid Fuel Cell MCFC – Molten Carbonate Fuel Cell DMFC – Direct Methanol Fuel Cell Note: Metal–air systems are often called metal–air batteries; they do not have a single universal acronym.
Quick Comparison Table
| Type | Operating Temperature | Typical Fuel | Typical Application |
|---|---|---|---|
| PEMFC | Low (60–80°C) | Hydrogen | Vehicles, backup power |
| SOFC | High (600–1000°C) | Hydrogen / Natural Gas | Grid & industrial power |
| AFC | Low (60–90°C) | Hydrogen | Space applications |
| PAFC | Medium (\~200°C) | Reformed Natural Gas | Commercial buildings |
| MCFC | High (\~650°C) | Natural Gas / Biogas | Large-scale power plants |
| DMFC | Low (50–120°C) | Methanol | Portable power devices |
| Metal–Air (e.g., Mg–air, Zn–air, Al–air) | Low (ambient) | Metal (Mg/Zn/Al) + oxygen from air | Primary batteries, specialty/field power, demos & research |
Chemical Reactions:
- Anode Reaction (Oxidation): Mg -> Mg2+ + 2e-
- Cathode Reaction (Reduction): ½O2 + H2O + 2e- -> 2OH-
- Cell Reaction: 2Mg + O2 + 2H2O -> 2Mg(OH)2
- Corrosion Reaction: Mg + 2H2O -> Mg(OH)2+H2
- The chloride ions (Cl-) in the electrolyte cause the anode to self corrode.
Did you know???
- Ancient Tech: Although they seem futuristic, the first fuel cell was invented way back in 1839 by Sir William Grove.
- Space Proven: NASA has used fuel cells since the 1960s to power Apollo missions, satellites, and space shuttles.
- Water Only: When run on pure hydrogen, fuel cells emit zero emissions at the point of use—only water vapor.
- No Moving Parts: Many fuel cell systems are stationary with no moving parts, making them extremely quiet and requiring minimal maintenance.
- Space Proven: NASA has used fuel cells since the 1960s to power Apollo missions, satellites, and space shuttles.
Variables
Controlled Variables
- Container size and material
- Volume of water (250 mL)
- Magnesium plate thickness and surface area
- Magnesium material (same sample type)
- Type of salt (sodium chloride, NaCl)
- Type of water (reverse osmosis water)
- Activated carbon cathode material
- Distance between electrodes
- Oxygen availability (passive air exposure)
- Ambient and electrolyte temperature
- External load resistance (220 ohms)
- Measurement equipment (same multimeter and setup)
Independent Variable
- Salt concentration in the electrolyte (0 g, 15 g, 30 g, 45 g dissolved in 250 mL of water)
Dependent Variables
- Voltage (V)
- Current (A)
- Power output (W = V × I)
Procedure
Materials:
- Magnesium plate (constant thickness (1.0mm) & surface area)
- Activated carbon (measured masses per trial: 15g 30g & 45g)
- Copper wire (stripped, 1 per cathode)
- Coffee filters (4 per cathode)
- Sodium chloride (NaCl)
- 2L Reverse osmosis (RO) water (allow to reach room temperature, 22OC)
- 250 mL measuring cup
- Digital scale (±0.1 g)
- Multimeter with leads and alligator clips
- Electrical tape
- Hot glue with hot glue gun
- Zip ties (2 per cathode)
- Ventilated fuel cell container (3D printed)
- Watch or Timer
- Electrical load circuit with on/off switch and 220 ohm resistor
Magnesium–Air Fuel Cell Assembly Procedure:
A) Anode Assembly (Magnesium Electrode)
-
Container Preparation
- Thoroughly rinse the fuel cell container to remove all residual electrolyte or corrosion products.
- Dry completely before assembly.
-
Install Magnesium Anode
- Insert the magnesium plate into the anode retaining slot.
- Ensure one corner protrudes externally to allow secure connection of the multimeter alligator clip.
- Confirm the immersed portion is fully inside the electrolyte chamber and not contacting the cathode assembly.
-
Seal the Anode Slot
- Apply hot glue to seal the retaining slot.
- Ensure complete perimeter sealing to prevent electrolyte leakage.
- Allow glue to fully cure/dry before proceeding.
-
Leak Test
- Fill the container with RO water to a level above the anode slot.
- Inspect visually for leaks at the sealed interface.
- If leakage is observed:
- Drain container
- Dry thoroughly
- Remove and reseal
- Repeat leak test
- Once confirmed leak-free, empty and dry the container.
B) Cathode Assembly (Air Cathode – Activated Carbon)
- Construct the Carbon Cathode Core
- Lay one coffee filter flat.
- Fold side edges inward to create a pocket.
- Add a measured amount of activated carbon (consistent mass for repeatability).
- Strip insulation from copper wire.
- Form the copper wire into a spiral or coil to increase contact area.
- Embed the copper spiral within the activated carbon (ensure good electrical contact).
- Fold and seal the open edge using electrical tape.
- Ensure one end of the copper wire protrudes externally for multimeter connection.
- Reinforce the Cathode
- Place the sealed cathode assembly inside a second coffee filter.
- Seal again with electrical tape to improve mechanical strength and prevent rupture.
- Create Electrolyte Wicking Layer
- Take two additional coffee filters.
- Overlap them and position so they extend downward into the electrolyte.
- These filters must:
- Dip into the electrolyte
- Maintain contact with the carbon cathode
- Allow capillary wicking of electrolyte upward to cathode
- Final Cathode Mounting
- Place the cathode assembly on the ventilated lid.
- Ensure:
- The activated carbon layer faces outward toward air.
- The wicking filters dip into the electrolyte when assembled.
- Secure the entire assembly using zip ties.
- Place the ventilated lid onto the fuel cell container.
- Cathode must not directly contact the magnesium anode.
- Copper conductor must not touch electrolyte except through carbon matrix.
- Air access to carbon layer must remain unobstructed.
C) Salt Water Electrolyte Preparation
- Prepare Electrolyte
- Measure 250 mL of RO water at room temperature.
- Weigh the desired mass of NaCl (e.g., 0 g, 15 g, 30 g, 45 g).
- Add salt gradually to the water.
- Stir until completely dissolved.
- Visually confirm no undissolved crystals remain.
D) Final Assembly & Testing
- Fill the fuel cell container with prepared electrolyte
- Install the cathode lid assembly
- Cathode must not directly contact the magnesium anode.
- Copper conductor must not touch electrolyte except through carbon matrix.
- Air access to carbon layer must remain unobstructed.
- Connect, ensure switch is in open position and load resister is 220ohms
- Magnesium plate → multimeter negative terminal
- Copper cathode lead → multimeter positive terminal
- Measure
- Open circuit voltage (OCV)
- Close switch and measure
- Load voltage (OCV) every 1 minute for 30 minutes
Observations
Observation of the chemical reaction: The water got cloudy during the experiment There were bubbles coming from the magnesium plate?
Fact or Fiction? Is pure water an insulator? I assumed pure water would not conduct and that I could use it as a control. I used water from my parent’s reverse osmosis system.
Research: Reverse Osmosis is a water purification process that uses a semi-permeable membrane to separate water molecules from other substances.
Research: Water undergoes autoionization in the liquid state when two water molecules form one hydroxide anion (OH−) and one hydronium cation (H3O+). Because of autoionization, at ambient temperatures pure liquid water has a similar intrinsic charge carrier concentration to the semiconductor germanium and an intrinsic charge carrier concentration three orders
Research: iSpring Reverse Osmosis system home. The core model RCC7AK reduces TDS (93-98%), PFAS (96-99%), Chlorine (95-99%), Asbestos (>99.7%), Barium (>98.4%), Cadmium (>98.8%), Chromium (>99.2%), Copper (>97.8%), Fluoride (>97.4%), Lead (>98.9%), Selenium (>99.0%), and over 1,000 other contaminants.
Research: When pure water is exposed to air it absorbs contaminants such as atmosphere gases. Carbon dioxide readily dissolves in water to produce carbonic acid which is slightly conductive.
Research: Water exposed to air absorbs carbon dioxide, which forms carbonic acid. Carbonic acid dissociates to form ions, which increase the conductivity of the water and thus increases its conductivity.
Observation: When the solution was pure water, the circuit conducted electricity. There was a very small amount of electricity. What does this tell me? I infer that the water must be contaminated. The water was clear but it must have still contained some ions.
Observation: The copper turned green in some sections, dying the coffee filter green as well. I infer that the salty water sped up the oxidisation process of the copper wire. In the long run copper would affect the performance of the fuel cell but for my experiment it’s not as noticeable.
Fuel Cell Principals Why does the magnesium plate dissolve? Salt (NaCl) separates when dissolved into (Na) sodium and (Cl) chloride. The chloride punctures through the protective film magnesium hydroxide dissolving the magnesium plate below.
Analysis
Was the Experiment Repeatable?
- Multiple trials at each salt concentration.
- The same materials and setup were used each time.
- Voltage and current trends were fairly consistent across trials.
Variability & Experimental Error
There were some variations observed between trials. Possible causes include:
- Slight differences in activated carbon packing
- Minor temperature changes
- Inconsistent spacing between anode and cathode
- Measurement uncertainty in the multimeter
- Leakage of electrolyte to alligator clip (see graph)
- The system was hand-built, introducing inconsistencies.
- Oxygen at cathode was not precisely controlled.
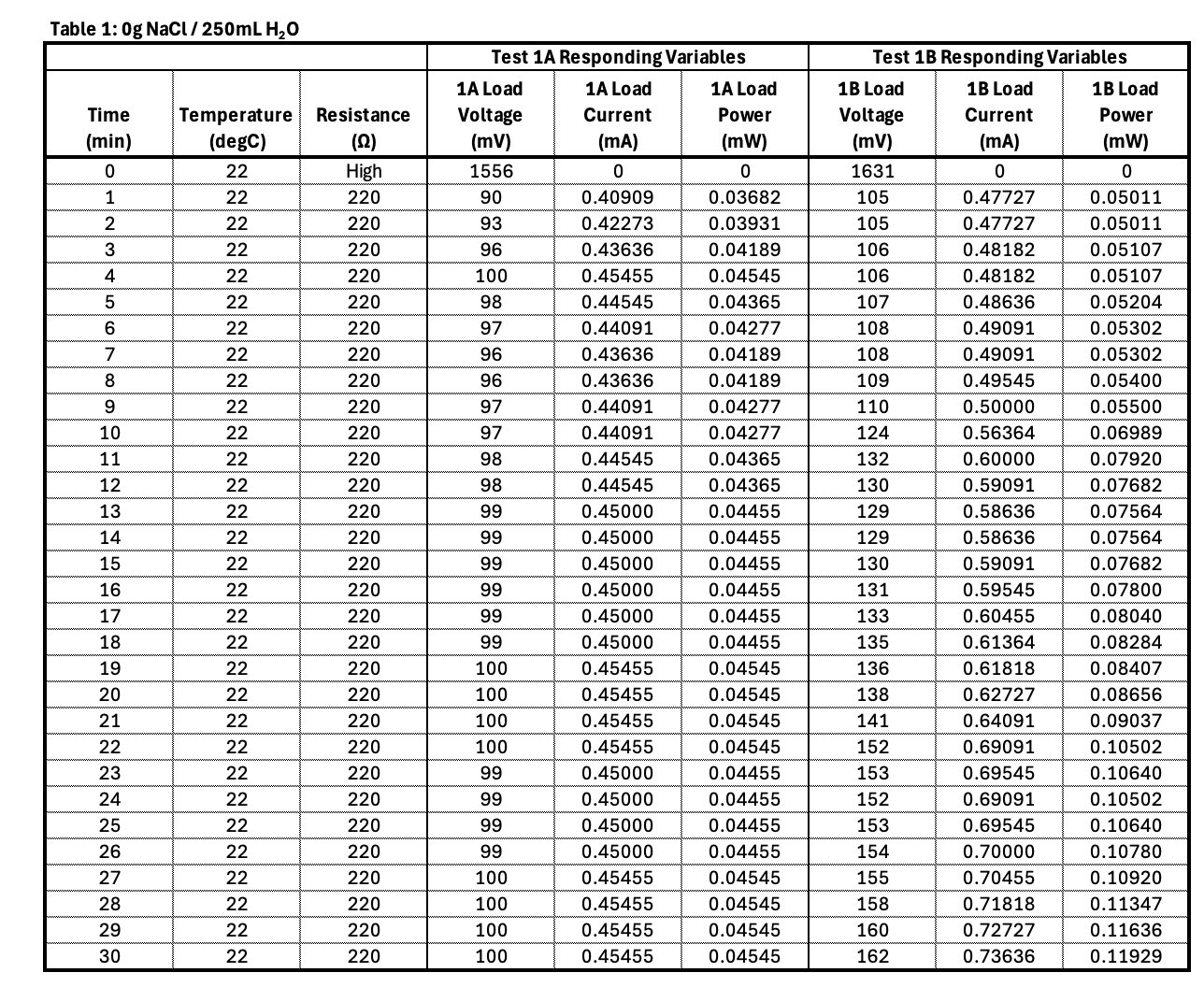
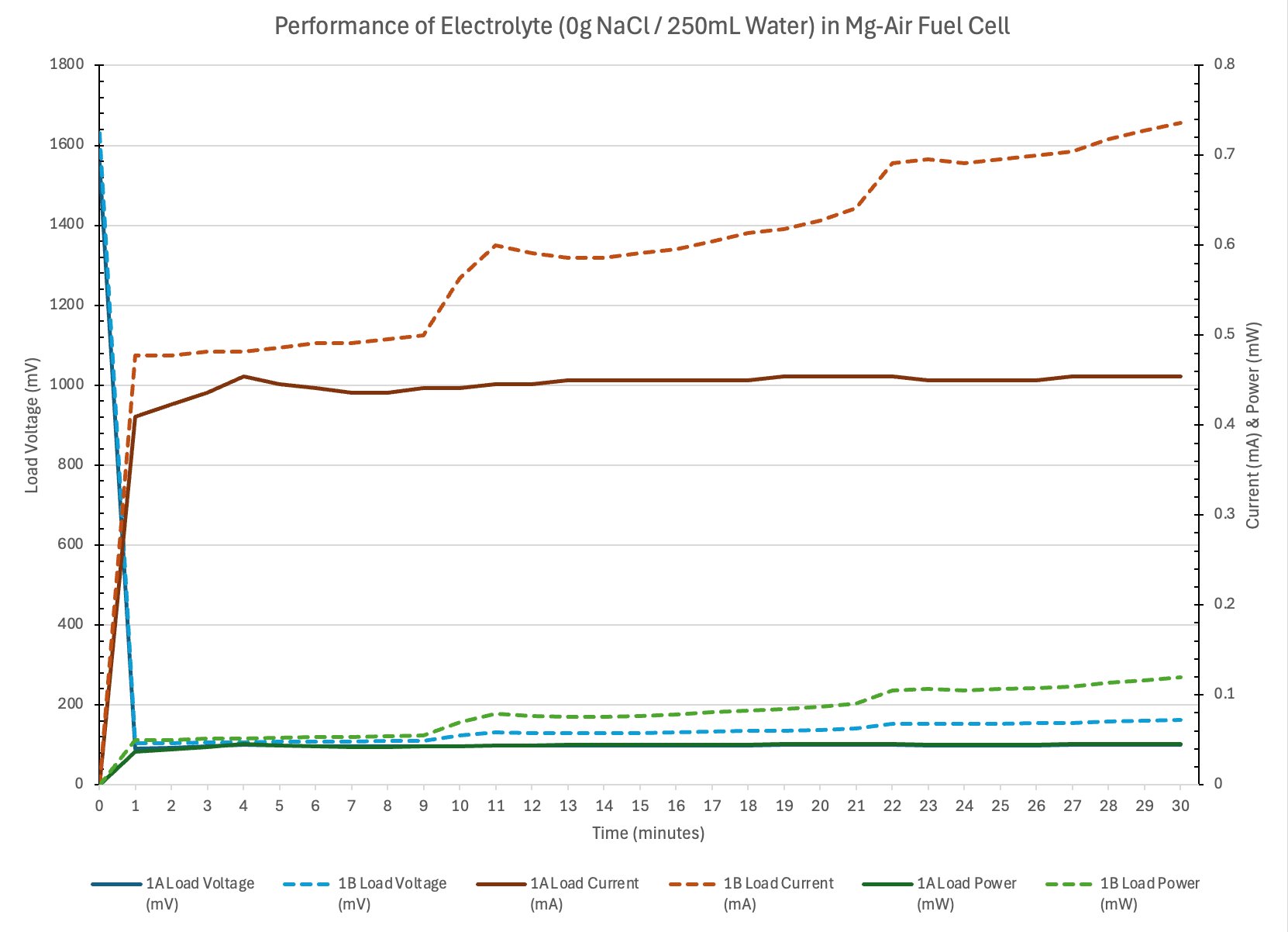
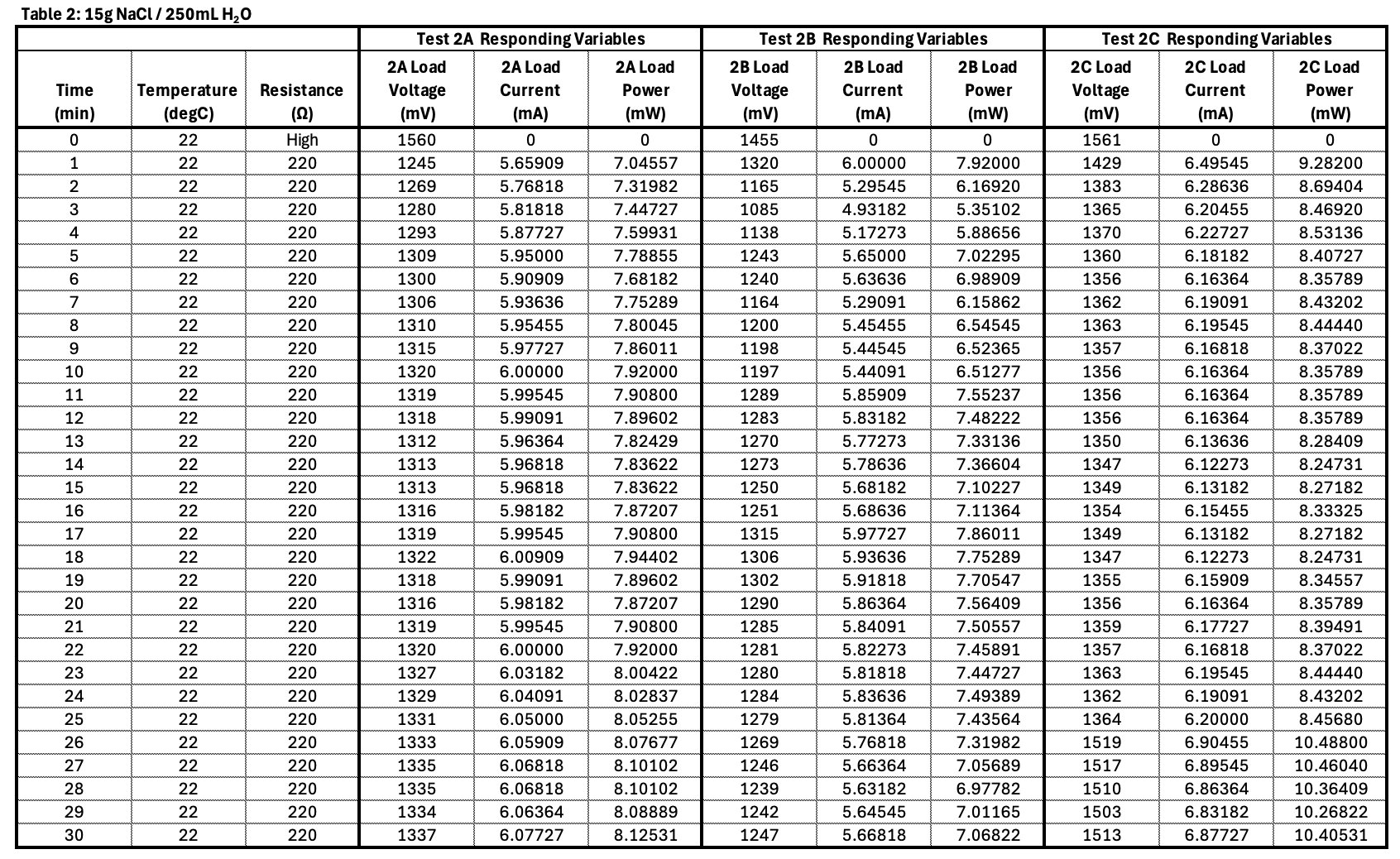
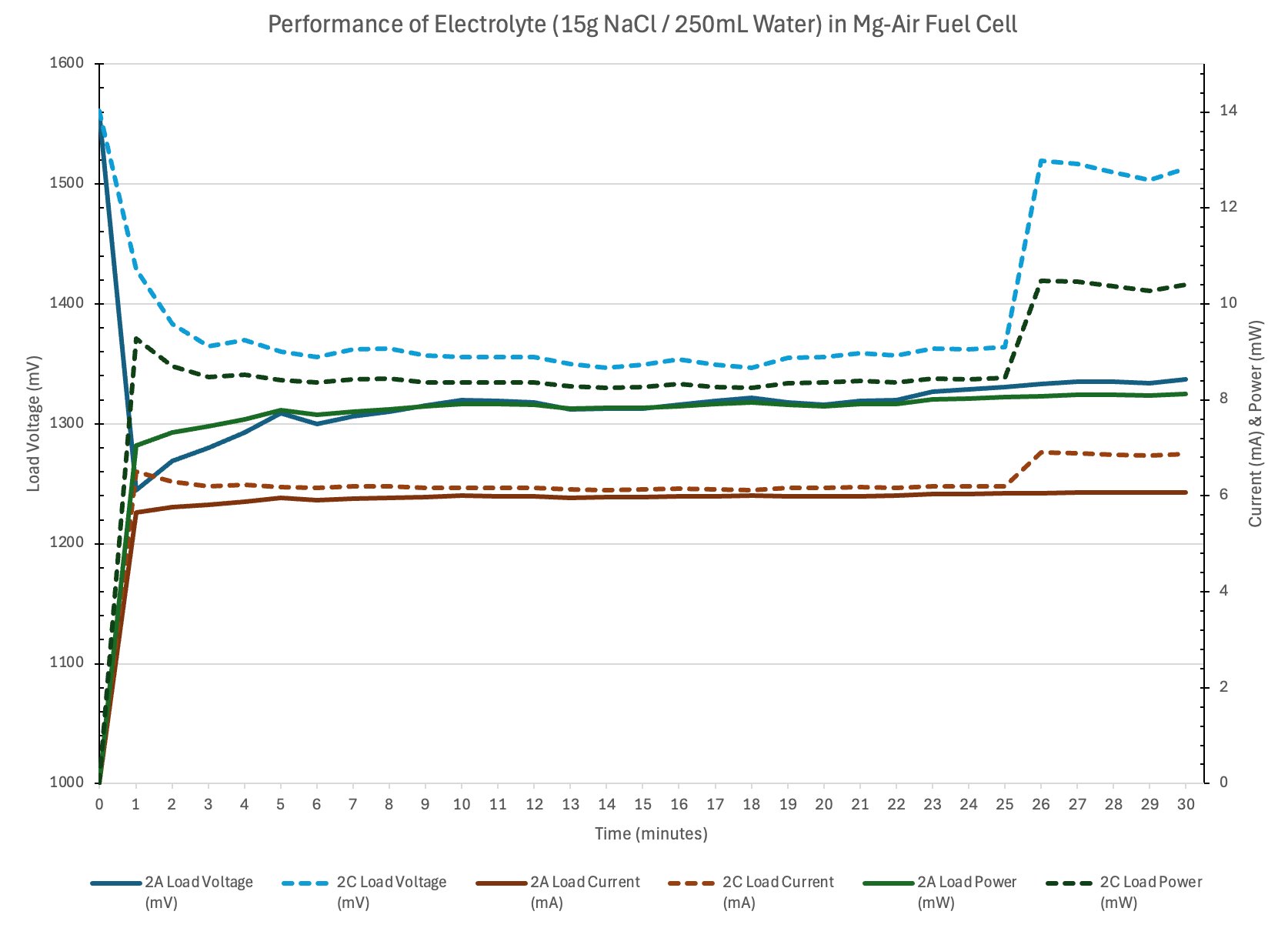
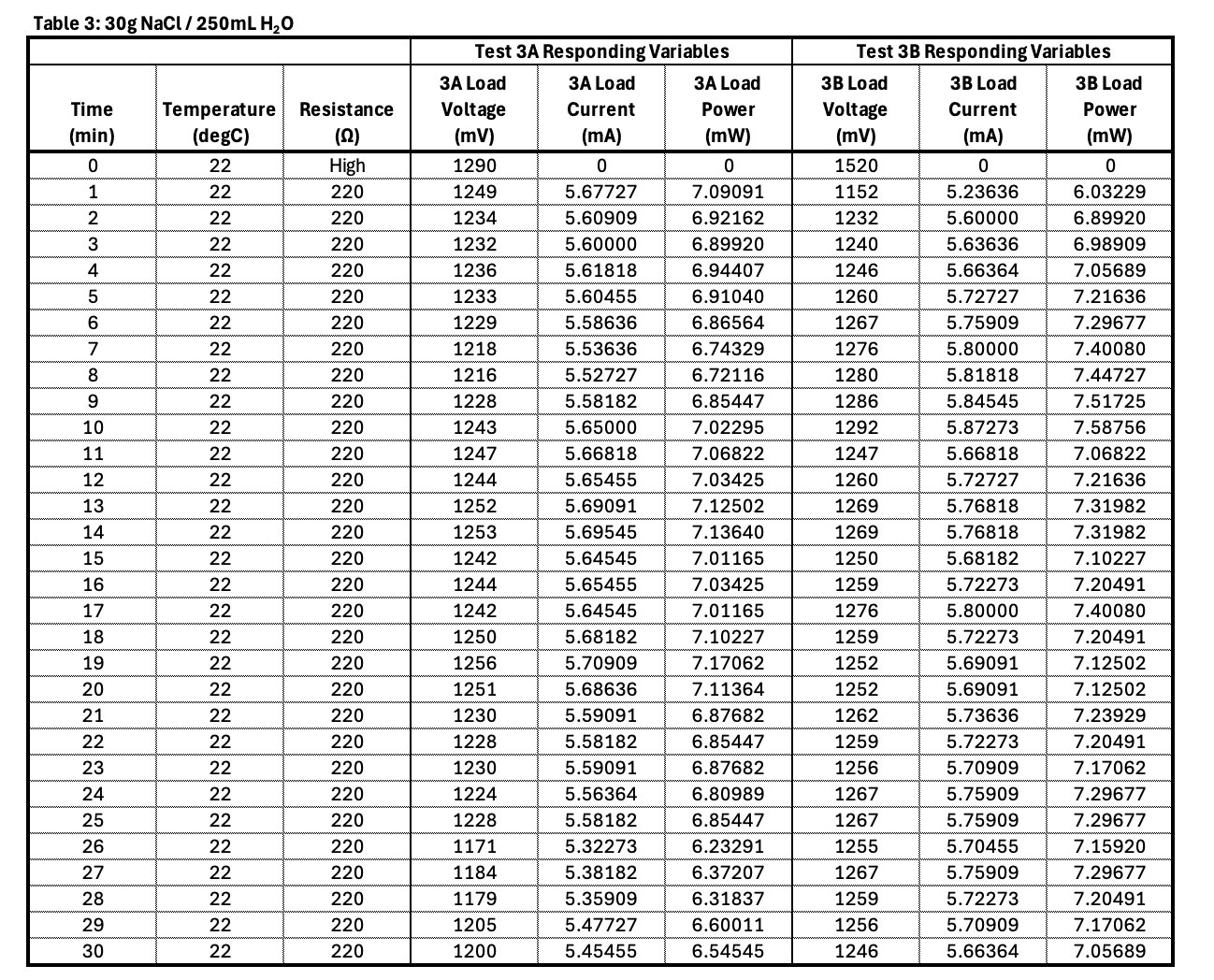
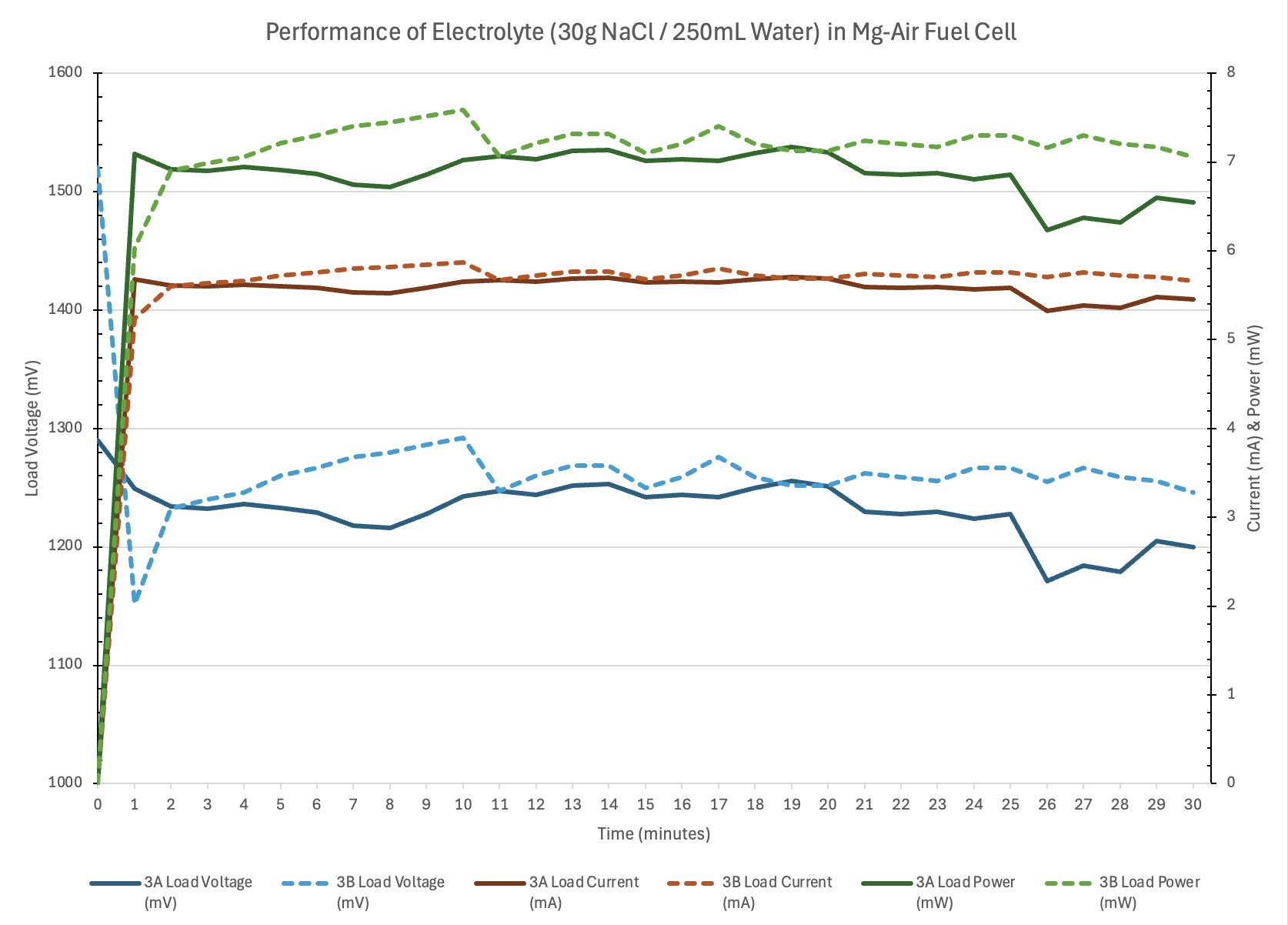
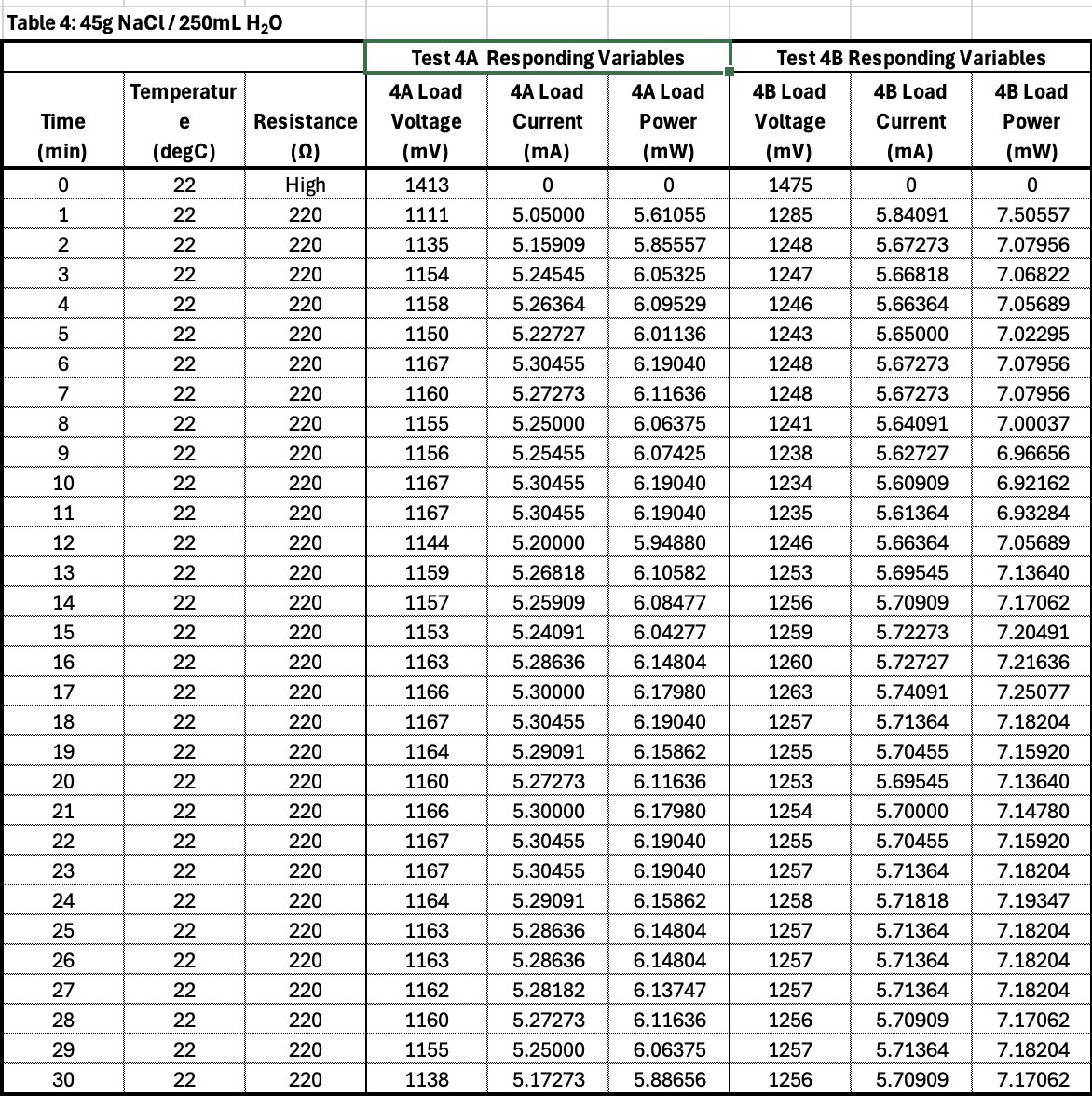
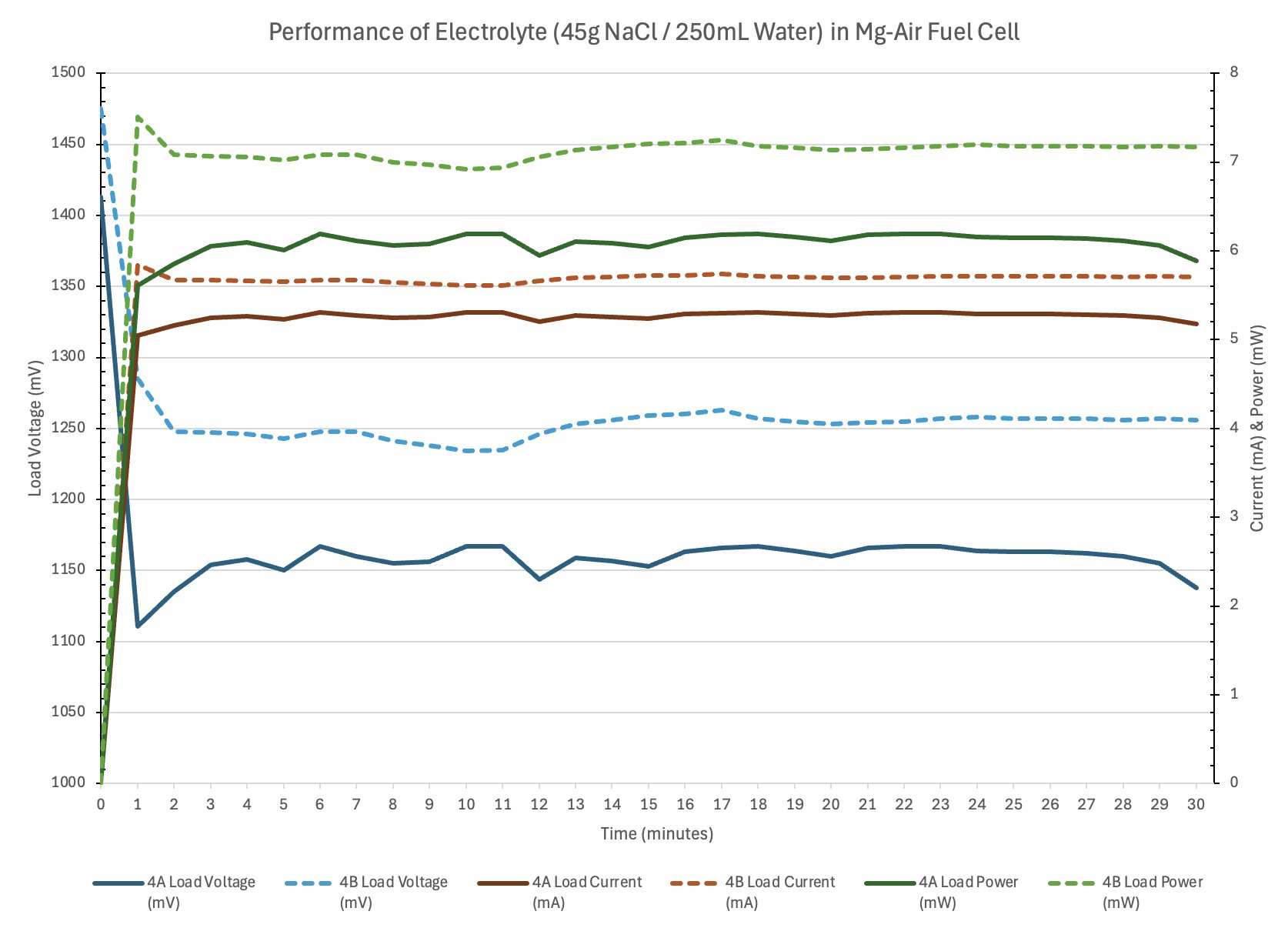
Conclusion
- Electrolyte concentration affects ionic conductivity.
- Too little salt means low conductivity and thus low current.
- Optimal concentration will produce adequate current with limited parasitic corrosion
Application
Real World Implication - Why do Fuel Cells Matter?
Fuel cells may help:
- Reduce greenhouse gas emissions
- Lower air pollution in cities
- Store renewable energy
- Provide clean backup power
What about Magnesium Air Fuel Cells?
Magnesium–air fuel cells (like this experiment) have potential for:
- Emergency power in remote areas
- Marine or underwater sensors
- Military and survival equipment
- Disposable high-energy batteries
Why Magnesium?
- Lightweight
- Abundant (8th most abundant element in crust)
- High energy density
- Uses oxygen from the air for reaction
However, challenges include:
- Corrosion in salt water
- Limited reusability
- Managing electrolyte concentration for optimal performance
Sources Of Error
- Salt weight not exact: Measuring salt using cooking scale may not be exact enough.
- Volume errors: Measuring using cooking measuring cups may change the volume of water a little between trials.
- Parasitic corrosion: may be occurring at different speeds for different salt concentrations.
- Copper wire corrosion: copper wire was observed to turn green. This might affect current flow.
- Alligator clip corrosion: in some trials, it was observed that even a little leakage of the salt solution onto the alligator clips caused them to react with the solution.
- Electrolyte leakage: the loss out of the container reduces the amount of electrolyte
Citations
- Youtube Salt Water Fuel Cell Demo Retrieved from https://www.youtube.com/watch?v=X9lRPgCfJ74
- Amazon.ca Magnesium Electroplating Experiment Machine Thickness Retrieved from https://www.amazon.ca/Magnesium-Electroplating-Experiment-Machine-Thickness/dp/B0C3YRTKJM?th=1
- Tinkercad from https://www.tinkercad.com
- Encyclopaedia Britannica Fuel cell Retrieved from https://www.britannica.com/technology/fuel-cell
- U.S. Department of Energy Comparison of fuel cell technologies Retrieved from https://www.energy.gov/eere/fuelcells/comparison-fuel-cell-technologies
- Phys.org (2010) Magnesium as an alternative power source Retrieved from https://phys.org/news/2010-04-magnesium-alternative-power-source.html
- Wikipedia Water fuel cell. Retrieved from https://en.wikipedia.org/wiki/Water_fuel_cell
-
UMICORE Fuel cells vs. batteries: key differences. Retrieved from https://www.umicore.com/en/media/newsroom/fuel-cells-battery-difference/
-
University of California, Santa Barbara Physics Lecture Demonstrations: 64.16. Retrieved from https://web.physics.ucsb.edu/~lecturedemonstrations/Composer/Pages/64.16.html
- SodaSense Does water go stale? Retrieved from https://sodasense.ca/blogs/bubbly-blog/does-water-go-stale
- Wikipedia Reverse osmosis. Retrieved from https://en.wikipedia.org/wiki/Reverse_osmosis
- Science Direct (2026) Magnesium air batteries. from: https://www.sciencedirect.com/science/article/pii/S2213956726000101
- Encyclopaedia Britannica (2024) Metal-air battery. Retrieved from https://www.britannica.com/technology/metal-air-battery
- U.S. Department of Energy. (n.d.). Fuel cells. Retrieved from https://www.energy.gov/eere/fuelcells/fuel-cells
Acknowledgement
- Ms. Melissa Perez - School science fair coordinator
- Mr David Joseph - Science lab teacher / science fair coordinator
- Big thank you to my Mom and Dad for helping and putting up with me over the last few busy months.

