The Science Behind Popping Boba; Small Spheres, Big Science
Tavnain Kaur
Dr. Gordon Higgins School
Grade 8
Presentation
No video provided
Hypothesis
If we change the best method suitable for spherification to different substances, such as water, chocolate ice cream, and Coca-Cola, then we will not observe a perfect sphere. For example, the chocolate ice cream already contains calcium, which makes it a thick jelly substance if we mix it with sodium alginate, but if we mix it with calcium chloride first, then give it a sodium alginate bath, it should form perfect sphere boba, which is also called the process of reverse spherification.
| c.no | Substance | Best method to use | Why? |
|---|---|---|---|
| 1 | Water | Direct spherification | Neutral pH, no calcium |
| 2 | Chocolate ice cream | Reverse spherification | Rich in fat, rich in calcium |
| 3 | Coca-Cola | Reverse spherification | Too acidic for direct spherification |
Research
BACKGROUND RESEARCH! QUESTION-1: What is molecular gastronomy? The transformation of food ingredients through innovative techniques to create unique flavours, textures, and presentations, etc., using scientific methods, is known as molecular gastronomy.
QUESTION-2: What is spherification? How is it used in molecular gastronomy? Spherification is a molecular gastronomy technique that transforms a liquid into small, gel-like spheres with a liquid center that pops in the mouth. It involves mixing a liquid with an ingredient such as sodium alginate, derived from seaweed, and then dropping the mixture into a calcium chloride bath. The two ingredients react to form a thin membrane around the liquid, creating the spheres.
QUESTION-3: What is the difference between direct and reverse spherification?
In direct spherification, sodium alginate is mixed into the liquid and dropped into a calcium bath, while in reverse spherification, calcium is added to the liquid and it is dropped into a sodium alginate bath. Direct spherification works best with simple, non-acidic and neutral PH. while the reverse spherification works better with calcium-rich or acidic foods.
QUESTION-4: What is a chemical reaction? A chemical reaction is a process where substances change into new substances with different properties. During a reaction, atoms rearrange and form a new solution. Signs of a chemical reaction can include bubbles, colour changes, temperature changes, and changes in the state of matter.
What is a solution? A solution is a homogeneous mixture, which contains a solute and a solvent. A solute is a substance that is mixed with the solvent, while the solvent does the dissolving. TOPIC: What is sodium alginate> 1. Where does sodium alginate come from? It comes from the walls of the brown algae. The seaweed is processed to extract the alginate, which is then turned into sodium alginate.
2. Why does it form gels when mixed with calcium?
Sodium alginate forms gels because it reacts with calcium ions. The calcium connects the long chains of alginate together, which creates a jelly-like substance.
3. How does sodium alginate behave in water? Sodium alginate dissolves in water to make a thick, smooth solution. The sodium ions separate, leaving the alginate molecules free to move until they meet calcium, which causes them to gel.
TOPIC: What is acidity (pH)> 1. What does pH mean? pH means the potential of hydrogen or the power of hydrogen. It shows us how many hydrogen ions are in a liquid. For reference –
- More hydrogen ions = more acidic = lower pH
- Less hydrogen ions = less acidic (more basic) = higher pH
2. Who invented it and why? The pH scale was invented by Søren Peter Lauritz Sørensen, a chemist from Denmark ( a country in Europe ), in 1909. The concept of the pH scale was the slow transition from the old colour-change tests for indicating the degree of acidity or basicity to electrical methods. He created it to help scientists measure and compare how acidic or basic liquids are in a simple way.
TOPIC: What is calcium lactate? 1. Calcium lactate is a food-safe calcium salt made from calcium and lactic acid. It dissolves in water and releases calcium ions (Ca²⁺). Calcium lactate is often preferred over calcium chloride because it does not have a bitter\, salty taste.
2. What do the calcium ions do in the reaction Calcium ions (Ca²⁺) turn the liquid alginate solution into a gel as sodium alginate is made of long chains of molecules that float on the surface when mixed in water. But when calcium ions are added, they attach to special parts of the alginate chains and link them together. As more calcium ions connect more chains, a network forms, and the liquid becomes a soft solid gel.
Variables
Manipulated Variable -
-
Kinds of spherifications:
-
Direct spherification
-
Reverse spherification
-
Type of substance:
-
Water
- Chocolate ice cream
- Coca-Cola
Responding Variable -
- Success of spherification: whether popping boba forms in spheres or not
- Shape of popping boba: how spherical it is, measured by the diameter ÷ height ratio
- Observations: texture, stability, and appearance of the boba
Controlled Variable -
- Amount of sodium alginate mixed in each solution
- Amount of calcium added
- Volume of each drop (boba)
- Stay time in calcium chloride bath (60 seconds)
- Temperature of solutions (cold)
- Measurement method (using graph paper, same procedure)
- Same equipment and lab conditions
Procedure
Procedures (for direct spherification): Step:1 make the calcium solution-
- Put 1 cup of water in a clean bowl
- Measure 2.0 grams of calcium chloride/calcium lactate
- Stir the solution using a spoon until it's dissolved completely.
Step:2 prepare the sodium alginate solution-
- Measure and pour ½ cup of cold testing food into the blender
- Add 2.0 grams of sodium alginate in it.
- Add another ½ cup of testing food in the blender
- Blend it until it forms a smooth lump-free solution
- Pour it in a clean bowl
Step:3 put the calcium chloride solution and the sodium alginate solution in the refrigerator for at least 1 hour or overnight to prevent the bubbles and the foam.
Step:4 grab the pH test strips and dip it in the cold food solution, match the strip to the pH scale to see its acidity or basicity. Record the data in the lab notebook.
Step:5 test if you can make the spheres-
- Take a syringe and suck up a small amount of the ¼ cup food solution.
- Prepare a bowl containing cold water
- Take the bowl containing the calcium chloride solution and release one drop of the sodium alginate solution into it.
- Put a timer for 60 seconds and observe.
- After 60 seconds, scoop the boba out of the bowl and put it in the bowl full of water, take it out after around 10 seconds.
Step:6 Observe its texture, stability and appearance and collect the data in the lab book.
Step:7 measure if the boba is formed as a sphere using a 1 mm graph paper. Record the data in the lab book.
Step:8 measure the height of the boba by placing the graph paper vertically straight, look from your eye level for the correct measurement. Record the data in the lab book.
Step:9 Take pictures of the experiment and the boba.
Procedures (for reverse spherification): Step:1 make the sodium alginate solution-
- Put 1 cup of water in a clean bowl
- Measure and put 2.0 grams of sodium alginate.
- Stir the solution using a spoon until it's dissolved completely.
If the testing food already contains more than 15-20 % of the calcium, skip step 2.
Step:2 prepare the food with the calcium chloride solution-
- Measure and pour ½ cup of cold testing food into the blender
- Add 2.0 grams of sodium alginate in it.
- Add another ½ cup of testing food in the blender
- Blend it until it forms a smooth lump-free solution
- Pour it in a clean bowl
Step:3 put the calcium chloride solution and the sodium alginate solution in the refrigerator for at least 1 hour or overnight to prevent the bubbles and the foam.
Step:4 grab the Ph test strips and dip it in the cold food solution, match the strip to the Ph scale to see its acidity or basicity. Record the data in the lab notebook.
Step:5 test if you can make the spheres-
- Take a syringe and suck up a small amount of the food solution.
- Prepare a bowl containing cold water
- Take the bowl containing the sodium alginate solution and release one drop of the calcium solution into it. Put a timer for 60 seconds and observe.
- After 60 seconds, scoop the boba out of the bowl and put it in the bowl full of water, take it out after around 10 seconds.
Step:6 Observe its texture, stability and appearance and collect the data in the lab book.
Step:7 measure if the boba is formed as a sphere using a 1 mm graph paper. Record the data in the lab book.
Step:8 measure the height of the boba by placing the graph paper vertically straight, look from your eye level for the correct measurement. Record the data in the lab book.
Step:9 Take pictures of the experiment and the boba.
Observations
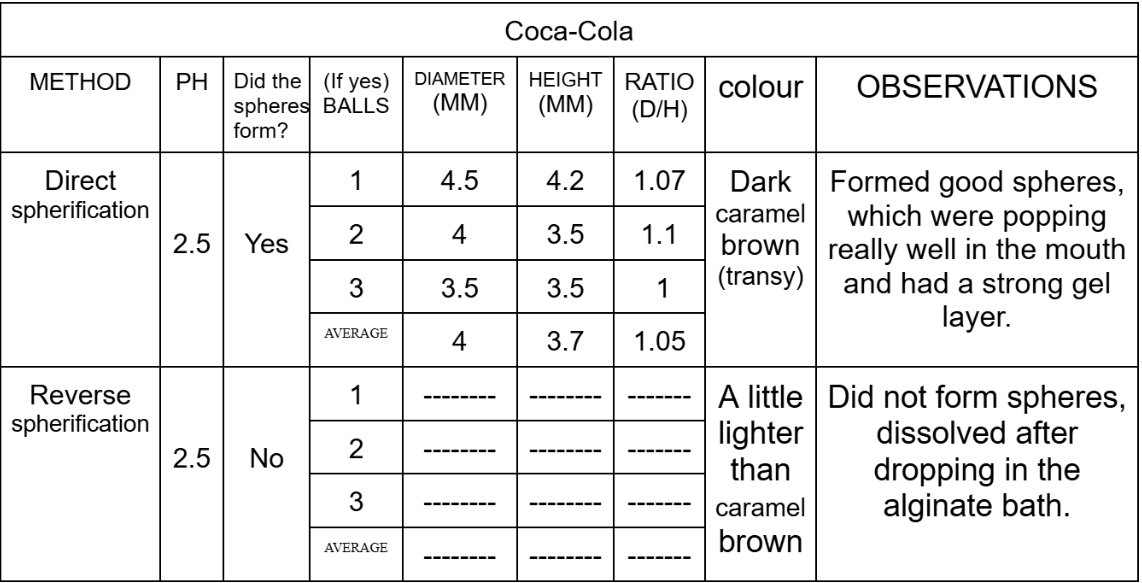
Size Differences of Boba in direct spherification:
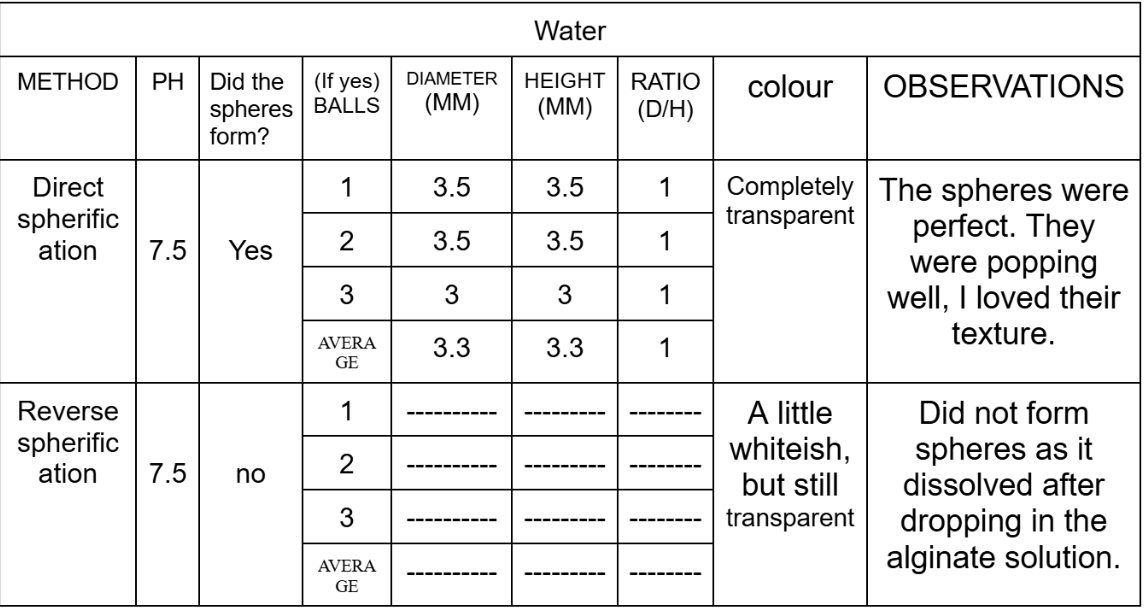
Size Differences of Boba in direct spherification:
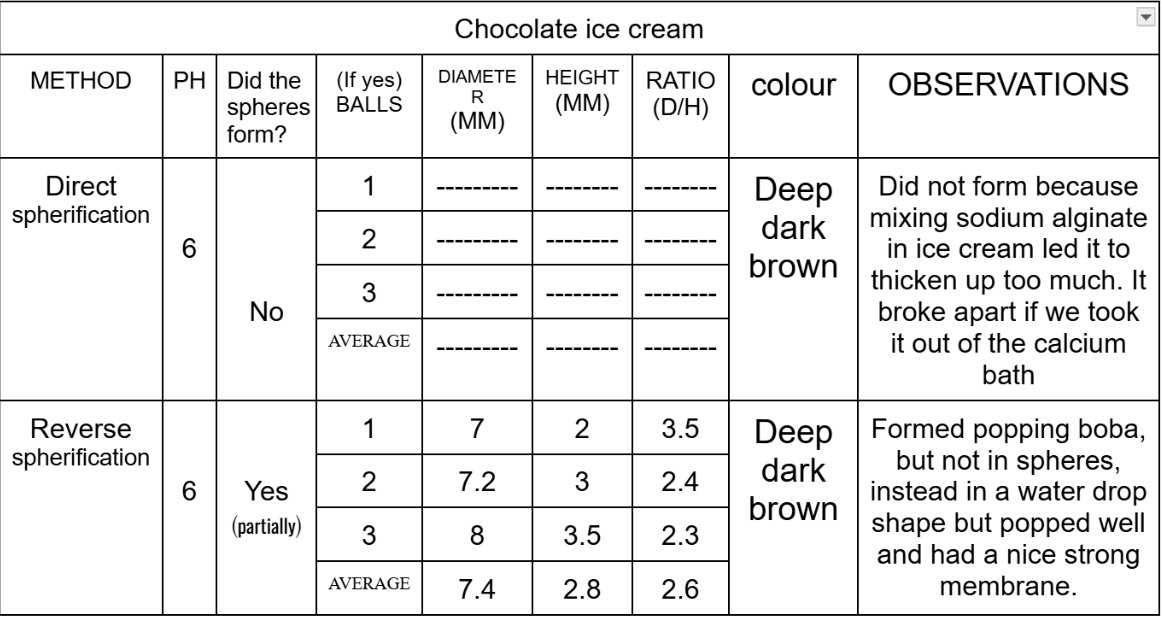
Size Differences of Boba in Reverse Spherification:
Additional observations: In direct spherification, the gelatinous layer formed on the inside, creating a jelly center in some spheres. In reverse spherification, the gelatinous layer forms on the outside, creating a shell that keeps the center liquid.
Analysis
After comparing the data, I have seen that all the different substances act differently to each type of spherification and have their own best-suited methods. Substances like water and Coca-Cola are thinner liquids and gets more thinner when we mix it with calcium chloride, so that is why it did not form spheres and just dissolved in the sodium alginate bath, while it worked best with direct spherification as when the sodium alginate was mixed with the water and the coke, it made the substance thick in consistency which helped it form perfect spheres. Substances with a concentration of calcium, such as the chocolate ice cream works best with reverse spherification as it forms a nice and strong gelatinous shell around the boba, while in direct spherification, it does not form a stable shell, leading to it melting as soon as we take it out of the calcium chloride bath.
Conclusion
The purpose of this experiment was to see how popping boba with substances like chocolate ice cream, Coca-Cola, and water using direct and reverse spherification affect the way the spheres form, and how pH influences which method works the best.
The results showed that each substance behaved differently depending on the spherification methods. The water only formed spheres in the direct spherification and dissolved in the reverse. The Coca-Cola formed spheres in the direct spherification although it was too acidic and dissolved in the alginate bath in the reverse spherification. The chocolate ice cream did not form spheres in any type of spherification although it still worked better with reverse spherification as it formed “drop” shaped boba, except like in the direct spherification where the boba melted as soon as we took it out of the calcium chloride bath.
The data of my experiment partially supports the hypothesis as it clearly stated that the best method to make popping boba using chocolate ice cream will be reverse spherification because of its thick consistency and richness in fat and calcium, and the results also show that the ice cream did not work in the direct spherification. However the hypothesis also stated that the best method of spherification for coke would be reverse spherification because of its low pH but it turned out to be direct spherification instead. This gave me a teaching that not only pH matters but the viscosity and the structure matters too.
This experiment shows that pH could affect spherification, but it also proves that it is not the only factor. Although Coca-Cola is acidic, it was still able to form stable spheres in direct spherification, while reverse spherification did not work well at all because the liquid was too thin. Overall, the results show that the success of spherification depends on a lot of factors such as pH, thickness, and ingredients, and choosing the correct method for each substance is important for forming stable popping boba.
Application
This experiment shows us that a variety of foods act differently if exposed to the same processes, as we saw for the direct and reverse spherification. Research on this topic is important in many different places, as food scientists and chefs need to know how different food ingredients or nutrients like fats, sugar, acidity, etc., affect the outcome’s texture and appearance. Even if one thing went wrong, or a measurement was incorrect, then the whole outcome could turn out dull, bitter and maybe the final output will not even form.
Understanding Molecular Gastronomy and the right type of spherification in relation to the substance’s pH and diversity of nutrients affects how well the spheres form. This experiment helps scientists figure out the correct method suitable for the best results.
Food scientists also need further research in this field, for more different Gelatinous cuisines such as yogurt, cheese, sauces, gummies/candies, or some specific drinks. A similar science is also used in medicines and pharmaceutical chemistry, where gels are used as ingredients in some kinds of medical drugs and sometimes used to protect some sort of sensitive ingredients.
Even though my experiment uses popping boba, it proves that understanding molecular gastronomy could be a necessity for future generations.
Sources Of Error
Some sources of error in this experiment could be the materials and tools used, as the pH strips used to measure acidity did not always give clear and exact results, which could have affected the accuracy of the recorded pH values. Small measurement errors could have occurred; the solution’s concentration and reaction time could also have affected the strength and shape of the spheres. Another source of error could be that the chemicals did not dissolve completely in the solution, which led to uneven and weak shell layers. Chocolate ice cream contains fat and air, which may have prevented it from forming in perfect spheres.
Citations
1. Cooking science\, boba spherification\, https://www.sciencebuddies.org/. Accessed 18 Nov. 2025
2. “Reverse Spherification.” Grokipedia\, https://grokipedia.com/page/reverse_spherification. Accessed 28 Feb. 2026.
3. “Alginic Acid.” Wikipedia\, 19 Feb. 2026. Wikipedia\, https://en.wikipedia.org/w/index.php?title=Alginic_acid&oldid=1339180471
4. O’Donnell\, Dominic. “What Does pH Stand For? Meaning\, Scale & Sensor Importance.” Sensorex Liquid Analysis Technology\, 11 Sept. 2023\, https://sensorex.com/what-does-ph-stand-for/.
5. What Is a Solution? https://www.edinformatics.com/math_science/solution.htm?fullweb=1. Accessed 28 Feb. 2026.
6. “Søren Sørensen.” Science History Institute\, https://www.sciencehistory.org/education/scientific-biographies/soren-sorensen/. Accessed 28 Feb. 2026.
7. Acids\, bases\, -pH. The pH Scale. Accessed 18 Nov. 2026
8. “Sodium Alginate.” Cape Crystal Brands\, https://www.capecrystalbrands.com/blogs/cape-crystal-brands/sodium-alginate. Accessed 28 Feb. 2026.
9. “Demystifying the Creation of Edible Spheres.” AIP\, 14 Mar. 2025\, https://doi.org/10.1063/10.0036245.
10. “The Impact of Spherification on the Gastronomic Experience.” Spherika\, https://caviarspherika.com/en/blogs/blogs/the-impact-of-spherification-on-gastronomic-experience. Accessed 28 Feb. 2026.
11. “The Spherificator® | Official Site.” The Spherificator® | Official Site\, https://thespherificator.com/. Accessed 17 Feb. 2026.
12. What Is a Solution? https://www.chem.purdue.edu/gchelp/solutions/whatis.html#:~:text=A%20solution%20is%20a%20homogeneous,distributed%20among%20the%20solvent%20particles. Accessed 28 Feb. 2026.
13. Abka-khajouei\, Roya\, et al. “Structures\, Properties and Applications of Alginates.” Marine Drugs\, vol. 20\, no. 6\, May 2022\, p. 364. PubMed Central\, https://doi.org/10.3390/md20060364.
14. PubChem. Sodium Alginate. https://pubchem.ncbi.nlm.nih.gov/compound/Sodium-Alginate. Accessed 28 Feb. 2026.
15. Colin. “What Is Sodium Alginate? | Sodium Alginate Applications | SNP Inc.” SNP\, 23 Apr. 2025\, https://www.snpinc.com/what-is-sodium-alginate/.
Acknowledgement
Acknowledgements: I would like to acknowledge my teachers, Manreet and Mr. Le, for their continuous guidance and feedback throughout the experiment, including my family, for gathering the materials and helping me in complicated steps for the success of this experiment. I would also like to acknowledge the online educational resources which helped me understand the hard concepts such as molecular gastronomy, spherification and pH.
I would like to acknowledge the following web pages for the pictures I used:
- Science buddies - https://www.sciencebuddies.org/
- Bubble tea empire - https://www.bubbleteaempire.co.za/
- Re Agent - https://www.reagent.co.uk/

