Which Fruit Juice will make the Best Spheres During Spherification
Kanwarpreet Sangha
Khalsa School Calgary Educational Foundation
Grade 6
Presentation
No video provided
Hypothesis
If we use five different juices to make popping bobas using Sodium alginate as gelling agent then the juice with the highest pH value will make best spheres. Lower pH of drinks prevent the sodium alginate from gelling properly because of higher concentration of hydrogen ions resulting in formation of non- symmetrical spheres.
Research
Spherification of liquids is a food science project to convert drinks into semi-solid balls called popping bobas that pop in your mouth. The popping boba balls made in this experiment are created due to a chemical reaction between sodium alginate and calcium chloride. Sodium alginate is an extract from brown algae and is a gelling agent. Sodium alginate has negatively charged alginate molecules and positively charged sodium ions. Sodium alginate creates a liquid solution when dissolved as sodium ions disassociate from the alginate molecules. Once the solution is dropped in the calcium chloride bath calcium alginate is formed. Doubly charged calcium ions can bind two different alginate molecules simultaneously and gel like substance is formed. pH of food is one of the main factors that effect the process of spherification. A lower pH means more hydrogen ions. Hydrogen ions are more reactive than calcium ions and interferes in the process of spherification. Viscosity, calcium in food and temperature of liquids are the other factors that effect the process of spherification in liquids. In this experiment I will test five different juices to test the effect of pH on the shape of spheres. Research related to ingredients shows that liquid needs to be cold otherwise sodium alginate may gel even before it is dispersed in chloride bath resulting in gel lumps, acidity of liquids cannot be too high, pH of liquids need to be above 3.6. If the liquid has calcium then free calcium ions from the liquid will react with the sodium alginate even before we drop it in the calcium bath. Distilled water is used in the calcium bath due to tap water having calcium molecules affecting the results. Using distilled water can get us the most accurate results in my experiment.
Variables
Independent Variable: pH of the fruit juices.
Dependent Variables : diameter and height of the spheres.
Controlled Variables : fruit juices quantity , sodium alginate and calcium chloride quantity, time to blend the solution together, time to keep the solution in the refrigerator, time the solution drops are kept in the bath.
Uncontrolled Variables : calcium in juices, pectin in orange and apple juice and the viscosity of the sodium alginate and fruit juice solution.
Procedure
First, measure 2 mg of sodium alginate and add it to one cup of first fruit juice to be tested in a blender. Blend it for 30 seconds shaking two to three times in between to get a homogenous solution. Second, put this solution in refrigerator for two hours to settle down. Third, measure 2 mg of calcium chloride and mix it properly in one cup of distilled water to prepare calcium bath. Fourth, remove the fruit juice and sodium alginate solution from the refrigerator. Test the pH value of the solution. Fifth, take some solution with the help of the syringe from the bottom layer so that it can only sucks the liquid part leaving foam at top and wipe the syringe to clean any excess liquid on sides. Next, carefully release solution only one drop at a time to calcium chloride bath and make few balls. Let those sit in solution for one minute. Remove these juice balls with the help of a strainer and measure the longest and shortest diameter of five balls for each trial. Repeat these steps to do five trials for each fruit juice.
Observations
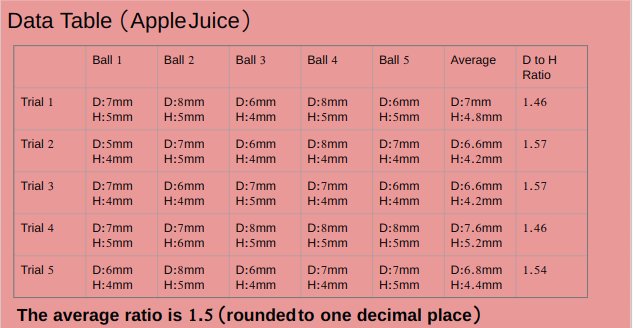
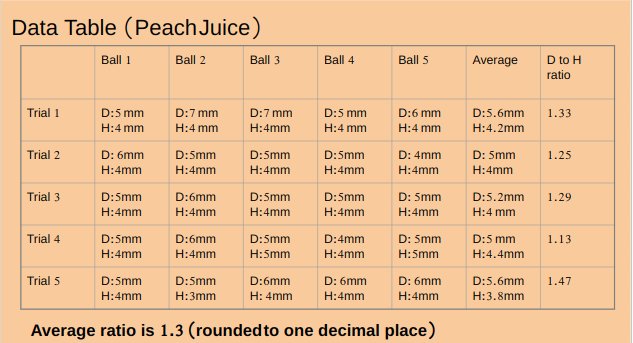
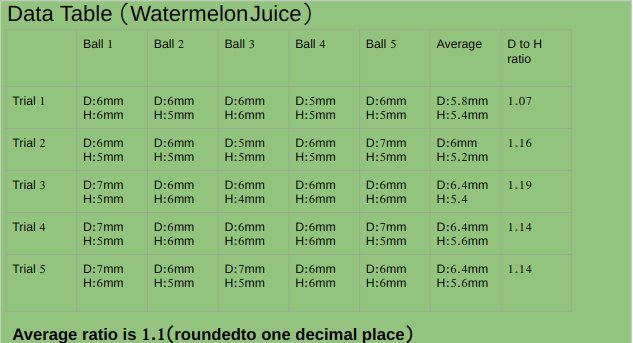
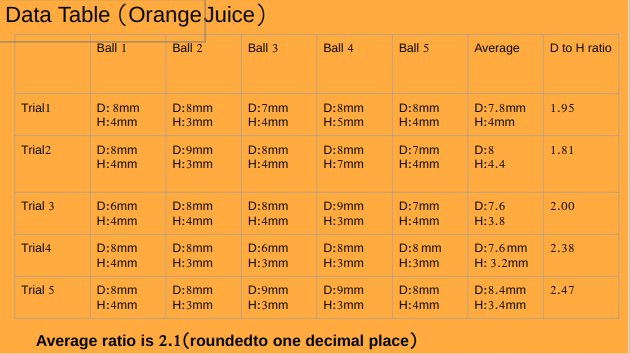
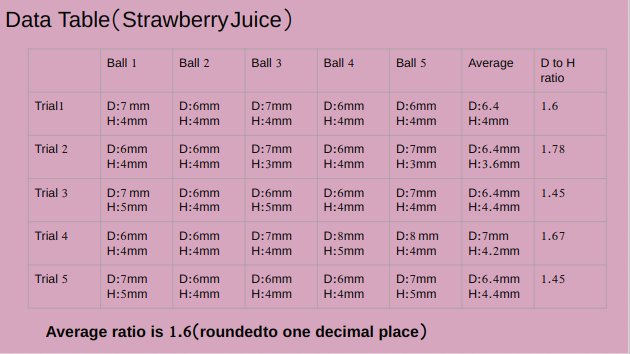
 pH value of apple juice is 4.16.
pH value of orange juice is 3.82
pH value of strawberry juice is 4.02
pH value of peach juice is 4.30
Ph value of watermelon juice is 5.07
Apple juice and alginate solution has a thick consistency.
Orange Juice and alginate solution has a very thick consistency
Strawberry juice and alginate solution has moderate consistency
Peach juice and alginate solution has moderate consistency.
Watermelon juice and alginate solution has thin consistency.
pH value of apple juice is 4.16.
pH value of orange juice is 3.82
pH value of strawberry juice is 4.02
pH value of peach juice is 4.30
Ph value of watermelon juice is 5.07
Apple juice and alginate solution has a thick consistency.
Orange Juice and alginate solution has a very thick consistency
Strawberry juice and alginate solution has moderate consistency
Peach juice and alginate solution has moderate consistency.
Watermelon juice and alginate solution has thin consistency.
Analysis
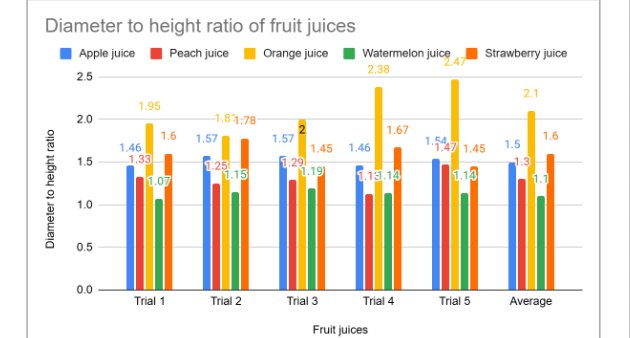
An experiment was conducted to test the effect of pH of liquids while making popping bobas from fruit juices. Five different juices Apple (pH 4.16),Peach ( pH 4.30),watermelon(pH 5.07),Orange (pH 3.82)and strawberry (pH 4.02) were tested. Watermelon juice with highest pH among all fruit juices made best spheres with average diameter to height ratio 1.1, followed by peach juice with average diameter to height ratio 1.3, followed by apple juice with average diameter to height ratio 1.5 , followed by strawberry juice with average diameter to height ratio 1.6 . Orange juice with least pH value made worst spheres with average diameter to height ratio of 2.1.Results supported the hypothesis of the project which stated that liquid with highest pH value will make best spheres.
Conclusion
The purpose of this experiment was to test which fruit juice from apple, peach, orange, watermelon and strawberry makes best spheres while making popping bobas. Solution of sodium alginate and fruit juice was dropped in calcium chloride bath to make spheres. Longest and shortest diameters of the spheres were measured and average of diameter to height ratio was calculated. Watermelon juice with highest pH among all fruit juices had a d/h ratio closest to one and made best spheres. It was hypothesized that as a result of this experiment the juice with highest pH will make best spheres. It is proved to be correct. Watermelon juice made best spheres as there were less hydrogen ions to block calcium ions from binding with alginate. Peach juice with second highest pH value got second best d/h ratio ,followed by apple juice which had third highest pH, followed by strawberry juice. Orange juice with lowest pH value made worst spheres.
Application
Fruit balls are used widely across the world in a vast variety of foods and drinks. Some popular uses of these fruit balls include bubble tea and frozen yogurt. Some other uses of these balls are in deserts such as ice cream, smoothies and milkshakes to add unique flavors to them. This is good for enhancing flavor because it could be used to correspond to what you are craving as these can be made with almost any fresh juices you like and add unique twist to your basic drinks and deserts.
Sources Of Error
There could be some measurement errors. When we put drops of fruit juice and alginate solution in chloride bath some drops stays little longer than others in the bath. This can effect the size of spheres. A better way is to put one drop at a time and measure it first . Uncontrolled variables like calcium and pectin in juices also effect the spherification poorly.
Citations
https://www.sciencebuddies.org/science-fair-projects/project-ideas/FoodSci_p074/cooking-food-science/boba-spherification https://www.sosa.cat/en/spherification/ https://blog.modernistpantry.com/advice/dropping-knowledge-in-sphere-form/#:~:text=Sodium%20hexametaphosphate%20is%20a%20sequestrant,pH%20to%20the%20proper%20levels https://specialingredients.co.uk/blogs/food-ingredients/how-to-make-caviar-pearls-using-spherification https://www.chefsteps.com/ingredients/distilled-water#:~:text=EDIT-,description,for%20cleaning/descaling%20the%20vessel https://www.epa.gov/goldkingmine/what-ph#:~:text=The%20pH%20scale%20measures%20how,greater%20than%207%20is%20basic https://itschemicallydelicious.wordpress.com/2013/02/07/the-science-behind-spherification/#:~:text=The%20second%20problem%20is%20when,an%20extended%20period%20of%20time
Acknowledgement
I acknowledge my school teachers Ms. Aulakh and Mrs. Sandhu for guiding me and I also acknowledge my mom to gather all the materials and support me during my experiment.

