What are you drinking?
Sukhnaaz Kaur
Annie Gale School
Grade 8
Presentation
No video provided
Hypothesis
If one cup of water is covered and the other is not, then the uncovered cup will have more dust particles and lower pH levels, making it more acidic than the covered cup because the cup exposed to air allows dust particles and objects to settle in the water (bacteria and germs), which makes it acidic.
Research
1. What is water PH?
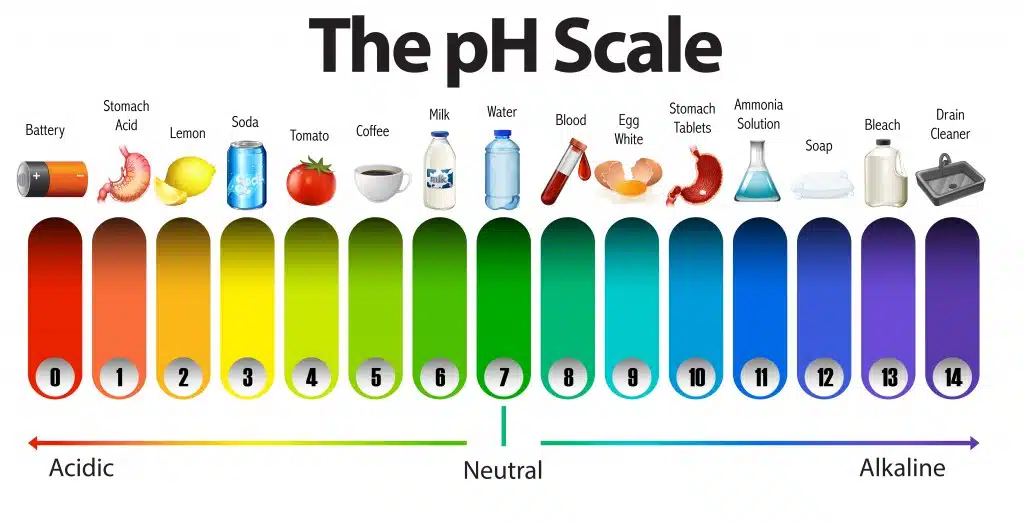
What does it mean to be acidic or alkaline? Why is the pH of water important to know? Ph indicates the acidity or alkalinity level of the
water. Acidic means the PH level is closer to 1, and alkaline means the PH level is closer to 14. It is essential to know water pH so that it
can be adjusted to a level that improves the health of people and environmental safety.
2. Does dust have anything to do with Ph levels in water?
Dust affects water pH. Dust particles, when they land in bodies of water, can act as either an acid or an alkali, altering the water's balance.
How it works:
Alkaline:
Soil dust containing calcium and magnesium carbonate. When this dust settles into water, it neutralises acid rain and raises the pH, increasing alkalinity.
Acidic:
Dust particles from industrial areas, such as kilns, ashes and dry deposits, can be highly acidic. When these settle in water, they can decrease the pH, making the water more acidic.
*Acid rain: Any form of precipitation with high acidity, usually below 5.6, caused by atmospheric pollution.
3. Can you make dust particles visible in water by dying them?
Dust particles are so small or transparent that staining helps increase contrast to make them easier to see.
- Methylene Blue (indigo-ish colour): It stains acidic cell parts and binds well to organic debris, turning them a deep blue.
- Iodine (brown colour): Excellent for staining carbohydrates and starch in plant-based dust (pollen, debris), causing them to turn brown or blue-black.
- Food Colouring: If you do not have laboratory stains, you can use blue or green food colouring to add contrast to the background and make transparent particles more visible.
- India Ink: While not a "dye" that changes colour, adding India ink to a water sample creates a dark background, making non-stained particles stand out through contrast.
4. Does dust have anything to do with ph level differences?
Yes, the pH level of water can change due to the settling of dust particles on its surface, particularly during events known as acid rain. Acid rain occurs when acidic dust and various other pollutants mix with water, leading to contamination that affects both the water and the surrounding environment. Once this polluted water enters the natural water cycle, it may precipitate as acidic rain, falling back to the ground and potentially harming the soil and local ecosystems. This process can contribute to soil quality decline and disrupt natural habitats, illustrating the significant environmental impact of changes in water pH caused by atmospheric pollution.
5. How to change ph levels in water?
Lowering pH Levels: Lemon Juice - Contains citric acid - Effective in decreasing pH - Used in culinary and cleaning applications Vinegar - Contains acetic acid - Helps reduce pH levels - Common household item
Raising pH Levels: Baking Soda - Chemical name: Sodium bicarbonate - Alkaline agent - Useful in cooking and neutralising acidity Cucumber Slices - Have an alkaline effect - Helps neutralise body acidity - Nutritious and hydrating option
pH Drops - Designed to alter pH levels - Used in gardening and aquariums - Ensures a balanced environment for plants and aquatic life
6. Should I be concerned about the ph level in water (side effects)?
If the water pH does not fall between the average (6.5 to 8), many side effects may show up over time.
If the water is highly acidic, it can contain lead, nickel, copper, arsenic and other materials. Side effects can include:
- diarrhea
- nausea and vomiting
- abdominal pain
- chills
- weakness
- shortness of breath
- suppression of the immune system
- organ damage
- Children show severe side effects from heavy metal exposure
- Development delays
- Respiratory issues
- Behavioural disorders,
- Certain forms of cancer
- Heart disease
Alkaline drinking water is considered safe. Currently, no evidence of negative side effects has been reported.
7. Types of dirt?
Many different types of dirt can have different types of risk. This is why I have categorised them as risk factors:
Low risk
- softwood dust
- soil
- house dust
- General Building waste
- solid surface materials
medium risk:
- hardwood dust
- MDF and man-made materials
- Cement and tile adhesives
- concrete dust
- quarterus materials
- oil and latex paints
high risk:
- Asbetos
- mold spores
- carcinogenic dust
- artificial mineral fibers
- bitumen
Combustible dust is a type of dust that is highly flammable. This type of dust is a very fine organic powder and, under the right atmospheric pressures, can be combustible.
8. Does pH affect taste?
When water has a pH below 6.5, it can sometimes taste a bit bitter. A lot of people don’t realise that this bitterness is linked to how acidic the water is. On the other side, if the pH goes above 8.5, it can taste similar to baking soda and leave a weird coating in your mouth.
This happens because pH affects the way carbonates behave in the water. When the pH changes, different forms of carbonates come to alter the chemistry of the water and also how it tastes and feels when you drink it.
So, in conclusion, pH is super important for how we experience the taste of water. It goes beyond just flavour; it affects our overall enjoyment and hydration experience. Keeping water in that sweet spot pH range can really make a difference in how good it tastes!
9. What might pH do to the environment (water bodies)?
Based on how acidic or alkaline the water is and what's in or around it, there are several effects that can happen. listed below.
Low pH
Effects:
- Damage to the gill epithelium
- Mucus on gills
- Decreased growth
- Reproductive failure
- Respiratory struggles
- Reduced the number of species
- death
- Change of species
High pH
Effects:
- Decreased reproduction
- Reduced biodiversity
- Decreased growth
- Damage to skin, gills, olfactory organs, and eyes.
Variables
Controlled:
- size of cups
- Which cups are covered
- amount of water(15ml)
- type of water purified
Manipulative:
- amount of dust fall
- type of dust
Responsive:
- ph level
- contaminatnts
Procedure

Materials:
- Two same-sized plastic cups ( transparent)
- Saran Wrap
- one rubber band
- One water bottle( purified)
- pH water strips
- measuring cup
- dropper
- microscope
- black marker
Procedure:
- Set out all the materials
- Take the measuring cup and measure one cup of water
- Pour the measured one cup of water into the cup
- With the black marker, mark the line where the water rests
- Set this cup aside
- Now, repeat the process with the second cup
- Place the Saran Wrap on the top of the glass
- Then, tie it in place with the rubber band
- Set both glasses in the same area
*After a week's wait *
- Using the dropper, make the slide to see through the microscope
- load the slide on the microscope
- Draw what you see through the tube
- Using the pH water testers, dip them in the glasses to test the waters
Now the project is done!!!!!
Observations
| DATE | DAY | UN-COVERED GLASS(left) and covered glass(right) |
|---|---|---|
| JAN/25 | 1 | |
| JAN/26 | 2 | |
| JAN/27 | 3 | |
| JAN/28 | 4 | |
| JAN/29 | 5 | |
| JAN/30 | 6 | |
| JAN/31 | 7 |
Analysis
| Type of water | uncovered cup | covered cup |
|---|---|---|
| pH levels | acidic (3) | neutral (7) |
| contaminants amount | medium-high amounts of microscopic dust and visible dust in 7 days. | non-negligible amounts of microscopic dust and visible dust in 7 days. |
| evaporation | 1.5 - 2 cm of water evaporation in 7 days. | No evaporation in 7 days. |
Conclusion
The goal of this experiment was to see if an uncovered cup of water would gather more dust particles and have a higher pH than a covered cup. I set up a simple experiment using two identical cups—one left uncovered and the other kept covered with Saran Wrap. Over a certain time, I gathered data on both cups to see how much dust was in the water and what the pH levels were.
After looking at the data, it was clear that the uncovered cup had a lot more dust particles. Plus, the pH levels in the uncovered cup were definitely lower than in the covered one. These results backed up my original hypothesis that covering the cup helps keep the water cleaner and safer to drink.
Based on these findings, there are a few interesting directions for future research. For instance, it would be intriguing to see how different types of glasses or lids affect contamination levels in the water. Also, examining how environmental factors such as location and how long the cups are exposed to air influence water safety could give us a better understanding of this issue. Overall, this study opens up new opportunities to look into better ways to treat and store water to improve public health.
Application
In my project, I learned a lot of things, from pH in water to different types of dust. This dust can affect drinking water; thousands of people don't have clean drinking water, not only because of pollutants like garbage, sewage, heavy metals, pesticides and fertilizers but also pH levels. Highly acidic pH levels can lead to death or severe problems. Other people have to walk long distances just to fetch a glass of two of water. that water is highly scarce in certain regions like the Sahara deserts and other dry places. In these places, it is important to conserve as much water as possible. For example, in Los Angeles, when the temperature rises, the water in water bodies starts to evaporate. In 2015, the Department of Water and Power in Los Angeles placed over 80 million black spheres in this body of water. When sunlight hits this body of water, it triggers a reaction between bromide and chlorine, which creates the carcinogen bromate (a potent oxidiser that damages DNA and causes nephrotoxicity). It also reduces up to 300 million gallons of evaporated water. These balls are called shade balls. The main ideology of my project is to thoroughly understand water contamination, Safe Drinking levels and water conservation, so the public can be informed on what they are drinking.
Sources Of Error
Some of the sources of error:
- When preparing slides, the slides got mixed up as I did not label them- solution- label them
- I noticed that both the cups were in the same area, but they did not get the same amount of sunlight because my window is at an angle- solution- place them closer to the window
Citations
Research
pic: https://aquaclearws.com/wp-content/uploads/2020/09/A-pH-scale-on-white-background-illustration-VectorBlueRingMedias-1.jpg.webp source: https://aquaclearws.com/resources/why-is-the-ph-of-water-important/
https://atlas-scientific.com/blog/what-causes-high-ph-in-pools/?srsltid=AfmBOopU96ed7Tw359A0Ttxq2wSn9dj9dwM3ozuRZvEVFChTlcww-PoC#:\~:text=Another%20environmental%20factor%20that%20can,contribute%20to%20a%20higher%20pH.
https://www.youtube.com/watch?v=C91BZsOb41o
https://www.healthline.com/nutrition/acidic-water https://www.healthline.com/health/food-nutrition/alkaline-water-benefits-risks#benefits
https://iwaponline.com/aqua/article/71/11/1278/91571/The-effect-of-pH-on-taste-and-odor-production-and
https://www.sciencedirect.com/science/article/abs/pii/S1352231013004706#:\~:text=Highlights,the%20occurrence%20of%20dust%20events.
https://www.wikihow.com/Lower-the-pH-of-Water https://www.epa.gov/caddis/ph#:\~:text=pH%20affects%20most%20chemical%20and,to%20aquatic%20plants%20and%20animals
Acknowledgement
I want to acknowledge my parents for helping me with my project. I would also like to appreciate the science teacher at our school, Mr Chiu, for his time and for allowing me to use the lab materials and classroom to test the water samples.

