Efficient Deicers: Comparing Acetates, Chlorides and Homemade Deicers
Jane Jang, Jimmy Xu
Queen Elizabeth High School
Grade 8
Presentation
No video provided
Hypothesis
General hypothesis:
If we test acetates, chlorides and homemade deicers, then calcium chloride would be the overall best for deicing sidewalks because calcium chloride is meant for fast-action deicing. It designed to work in temperatures below -15 degrees Celsius and less corrosive and more eco-friendly. Still, this is not fully the best deicer for every situation. This is because each deicer has pros and cons. For example, rock salt is very common, easy to access and is able to be used in many situations, but the brine creates is corrosive and harmful to our environment. We think that deicers should be used for its main purpose instead of a four in one deicing solution because of how hard it is to create the "perfect" deicer that can be used for all situations. We also think that deicers should be paired with additional support for snow and ice removal such as a snow plows/shovels and pebbles.
Chloride, Acetate and Homemade deicer hypothesis:
If you only wanted to use chlorides for deicing your sidewalks, Then calcium chloride would be better than rock salt because calcium chloride not only melts faster, it is more non-corrosive, and more efficient.
If you only wanted to use acetates for deicing your sidewalks, then calcium magnesium acetate would be overall better for deicing sidewalks because calcium magnesium acetate is eco-friendly and not corrosive. It also won't let the acetate brine refreeze as fast as other deicers.
If you only wanted to use homemade deicers for deicing your sidewalks, then the alcohol and soap mix would be better because it contains isopropyl alcohol/rubbing alcohol which is a really good deicer and the warm water and soap would allow immediate action and help spread the deicer equally. The alcohol water mix has too much water which ends up doing the opposite of what it is supposed to do. However, rubbing alcohol is highly flammable and not eco friendly so you have to make sure that you are adding the right ratio of water to rubbing alcohol.
Research
Background Research
What are deicers? Deicers are what keeps our important roads ice-free in the winter. These small grains of salt are what makes large roads less slippery and easier to drive. They are used in windshield fluid, sidewalk deicing, roads and planes etc. These help clear the snow and prevent snow accumulation. However they come with drawbacks, as most deicers have pros and cons. Take for example, rock salt, it is versatile, cheap and common, however, it is corrosive, harmful to our environment and ineffective below -9 Celsius.
Common ones you will find in a hardware shop include: -Calcium chloride -CMA/calcium magnesium acetate -Sodium chloride -Liquid deicers
How can we create a deicer that outclasses all the other deicers?
The main points in creating a deicer that works; a chemical that disrupts the lattice ice formation.however, creating one that is functional in every temperature and situation is challenging. For example, rock salt, it is common and cheap, but is unreliable below -9 Celsius and harmful to our environment. There really isn’t the “best” device that you can find and even if you did, it would probably be expensive. The main way to make a “good” deicer requires a blend of chemicals. An example of this is covering rock salt, a chloride that can disrupt the lattice formation of ice, with a corrosion inhibitor that can also disrupt the ice formation like CMA (calcium magnesium acetate) and urea, a commonly found natural chemical that your liver produces.
What makes deicers unsafe and non-environmentally friendly?
Most deicers either use acetates or chlorides, but both are harmful to our environment. Though acetates are mainly based on eco-friendly and safeness qualities rather than dissolving qualities, chlorides are rather irritating to handle. Chlorides are generally better at dissolving ice and snow but used without gloves during handling can cause skin irritation. Other crucial problems with chlorides is that they dry paws quickly for pets which can make them uncomfortable walking and even sometimes painful. Chlorides also kill plants quickly. This is why many people stick to shovelling or using natural deicers like CMA.
How do deicers actually “melt” ice and snow?
To clarify, deicers do not melt ice and snow like how you melt ice in a pan. If they did then you would see steam getting emitted. The actual “melting” is called freezing point depression, this process is dissolving rather than melting. However, there are some deicers that generate an exothermic reaction when in contact in snow such as calcium chloride. Another clarification is that salt doesn’t completely melt the ice and snow, it creates divots and holes in the ice and snow. This creates gaps in the ice which allows us to easily shovel it away.
Freezing point depression happens as chemicals interfere with the water molecules. The chemicals then dissolve and disperse throughout the ice. The chemical then mixes with the water, then disperse the water molecules which makes it hard for them to form the crystalline ice structure. Most of these chemicals are usually ions such as chlorides and acetates etc.
Process of freezing point depression: At 0 degrees Celsius, water molecules form a rigid and ordered hexagonal lattice structure (ice) through hydrogen bonding. Hydrogen bonding is when liquid water molecules that are constantly moving, break and reform, but when temperatures drop, the thermal energy escapes and the water molecules slow and then turn into ice.
When salt or a deicer is applied to the ice, ionic dissociation takes place. This process of dissociation creates particles that disrupt the crystal lattice of ice which turns it back into a liquid phase. A regular salt particle dissociates into 2 ions (Na+ , C1- )but calcium chloride releases 3 ions (Ca2+, C1- ,C1-) . Some examples of organic deicers are acetates and glycols.
What is refreeze and why it happens: Refreeze is when the brine (the dissolved liquid) refreezes because the deicer can't keep it liquid. The first reason why brine can refreeze into ice is because extremely low temperatures provide enough energy for the water molecules to freeze. This overcomes the salt ions and pushes them out which is called brine rejection. Another reason why brine freezes is because it becomes too diluted and the salt ions disperse out which allows the water molecules to freeze. The graph below shows different types of deicers and their recommended temperature use.
Another reason why deicers have a limit is because of eutectic temperatures. Each deicing agent has an eutectic temperature. This point is the lowest temperature possible where a chemical can keep water in a liquid state. When temperatures drop below the eutectic temperature, then the kinetic energy drops and turns the water into ice. Hygroscopic and exothermic limitations also occur. Calcium chloride and other chlorides need moisture from the air to start the freezing point depression. As the temperature drops, so does the moisture. Since calcium chloride also is an exothermic deicer, as soon as the ambient temperature (the actual measured temperature) drops too low, then the exothermic reaction becomes insufficient. Another limitation is the saturation limit. When the temperature drops extremely low, the water becomes saturated with the salt and cannot dissolve and continue the brining process. If the ambient temperature is lower than the saturation temperature, the brine freezes and the deicer cannot melt the ice further.
What affects a deicers performace?
Main things that affect de-icers:
Type of surface: Concrete and asphalt are different types of road surfaces. Asphalt is flexible and is darker compared to concrete. This means that it can retain the solar heat. Asphalt also has a higher thermal conductivity than concrete meaning it can transfer heat faster. It is also more corrosion resistant than concrete. Concrete however is light and can reflect the light onto ice. The type of surface material is also not the only thing contributing to ice formation. A porous surface attracts brine like a sponge and helps the re-freezing cycle. The porous surface is also more efficient than an impermeable surface because it allows the water to seep into the surface, lowering ice formation by 3 times.
Humidity: Air that has a higher humidity will have a higher heat capacity than dry. This difference is low but it still affects how ice melts. Colder days however will have less humidity than a warm day.
Wind: Chinooks or just regular above freezing wind can increase ice melting. This is because it increases the convective heat transfer. However, if it is below freezing, then it speeds up ice formation. Basically, wind speeds up freezing or melting depending on the temperature
Shape of deicer: Flakes, unlike clumps, don't get flung around by cars. Some cities use gravel for traction and by the end of winter when all the ice is gone, you will see gravel on the side ready to be swept up for another use. If we were to use clumps of salt, then the ice on the sides of the roads would be gone instead of the middle.
What's common to find in a deicing mixture and each chemical used in deicing. Usually, a deicer sold in an auto part or hardware shop would have a base chemical with fillers. It would be hard to find a 100 percent calcium chloride deicing mixture. A 99.5 percent of the main chemical would basically be the maximum for deicing. The place best for ordering 100 percent of the chemical you want is the very place it is created, like an industrial factory. A lot of deicers usually contain a mix of chemicals to improve the overall quality of the deicer. For example, a calcium chloride deicer would have calcium chloride as its base chemical, sodium chloride would be a filler. Other fillers could be urea (natural chemical found in our livers), abrasives like gravel, agricultural by-products to coat road salt, corrosion inhibitors, colorants, anti-caking agents to help spread the deicing agent and moisture inhibitors.
Other chemicals (deicing agents): A mix of chemicals like rock salt and magnesium chloride would benefit the deicing speed and efficiency. It helps give your deicing agent more uses and gives a better overall mix of chemicals
Corrosion Inhibitors: Urea, CMA (calcium magnesium chloride) and polyhydroxy carboxylic acid are corrosion inhibitors. They are usually labelled as enhancers on a deicing product but can actually be the main chemical. These chemicals prevent corrosion or minimize the effects of corrosion. They form a thin protective layer around the metal. This film neutralizes corrosive agents like chlorides, reduces oxidation or alters the electrochemical reaction. Types of corrosion inhibitors include: Anodic inhibitors: protect the anodic parts of the metal surface from oxidation Cathodic inhibitors: protect the cathodic parts of the metal surface from corrosion Mixed inhibitors: protects both cathodic and anodic Volatile corrosion inhibitors: These inhibitors protect long uses of metal items such as stored machinery.
Agricultural by-products An example of an ABP (agricultural by-product) is beet juice of molasses. They enhance performance of salt-based deicer, reduce operations costs, increase longevity, improve corrosion resistance and environmental impacts and corrosion resistance. These bio-based additives act as corrosion inhibitors, cryoprotectants and binding agents.
Abrasives: Chemicals break the ice apart, the abrasives help with traction and help prevent slipping and skidding. A commonly used abrasive is gravel. A down side to abrasives is that they can be easily kicked around and end up not on the sidewalks but on the lawn.
Colorants: Colorants help us see where we sprinkled our deicer. It also helps us see where we forgot to sprinkle the deicer or where we sprinkled too much deicer. It also helps with safety assurance so that dangerous areas like stairs and parking lots get covered in deicing agents.
Anti-caking agents: Anticaking agents help with storage, which prevent them turning into an unusable solid block. It helps even application, reduced wastage (applying too much) and increased flowability which prevents clumping.
Moisture inhibitors: Help with clumping issues, ice formation in the deicer, corrosion protection and improving adhesion.
Which one is better; a solid deicer or a liquid deicer?
A solid deicer is better after it snows but a liquid deicer is better before it snows. The type of deicer you use is highly dependent on the situation.
Overall, the type of deicer you use does not have a big impact. Both deicers still do the same job. Generally, solid deicers are more common as they were earlier to enter the market, however, liquid deicers are gaining popularity and are becoming more common and applicable for more uses.
How to maximize efficiency of your deicer?
The first way to increase your efficiency is to use the right amount of deicing agent. Using too little of your deicer minimizes the effect of the deicer. Using too much on the other hand is a waste of deicer and could be counterproductive. Once there is enough brine sitting on the top of the ice, there is no more room for more brine, so if you used tons of deicing agent, then a lot of the salt would sit at the top with no added benefit. By the time the ice has been dissolved by the brine, the leftover deicer is going to seep into the concrete and dry it until it cracks. Another way to maximize efficiency is pre-wetting rock salt with a liquid deicer like calcium chloride helps increase efficiency of your deicer.
What's the right amount of deicer to use? A good amount is 12 ounces of deicer for 250 square feet of pavement. If you are unsure, then you should check how far each particle of salt is to each other. A 1-3 inch space between each salt granule is good as long there are none stacking on each other. It is still best to check the instructions manual on the deicer container/bag/bottle because each deicer is different so the directors manual would have the most accurate results.
What makes deciders different from each other? Each deicer is different, however, they use the same method. So why do they perform differently? Each chemical interacts differently with ice during the lowering of the freeze point. They also have a different speed of dissolution (kinetics), endothermic or exothermic reaction and if they are hygroscopic are not.
-Magnesium chloride versus calcium chloride. What makes them different? Dry Cac12 flakes or pellets normally contain 77-83 percent active chemicals (deicing chemical) where as Mgc1+2 usually is often a hexahydrate (Chemical compound that contain 6 molecules of water of crystallization bonded into a crystal structure) meaning it's diluted with water, which means its 47 percent active chemicals Cac12 flakes or pellets are exothermic, which means they release heat as they dissolve the ice. This further accelerates the dissolving of the ice. Mgc1+2 is also exothermic but it is not as strong as Cac12 flakes or pellets exothermic reaction. This is because Mgc1+2 is smaller than Cac12 so it holds onto its chloride ion tighter. However, Mgc1+2 has higher hydration energy (dissolving the ice) but its high lattice energy traps more of the hydration energy then giving it off. The combination of lattice energy and hydration enthalpy makes Cac12 superior to Mgc1+2. However, Mgc1+2 has a higher charge density, leading to very strong hydration, but this often turns it into a hexahydrate, which means it already has water so it further reduces the heat released during dissolution. In conclusion, while magnesium attracts more water, the overall energy is less than Cac12. Using a anhydrous magnesium chloride would result in higher exothermic reactions than a hexahydrate and keep its high charge density but it still won’t beat calcium chloride’s high energy output. Dry Cac12 flakes or pellets are also more hygroscopic which means it attracts moisture which allows it to dissolve ice faster than Mgc1+2 .
Extreme deicing: glycols and isopropyl alcohol
Chlorides and acetates are not the best deicer, in fact, calcium chloride’s maximum eutectic temperature barely nicks the halfway mark for isopropyl alcohol.
There is also ethanol and methanol that also dissolves ice but they are toxic and should be avoided. These glycols are mostly used in planes.
How does isopropyl alcohol and glycols prevent ice formation?
Isopropyl alcohol and glycols are polar liquids. Polar liquids consist of a net dipole moment, meaning they have distinct , slight positive and negative ends caused by electronegativity differences and asymmetric molecular shapes. Polar liquids dissolve ice primarily by introducing energy to break the hydrogen bonds holding the ice structure together. Because ice is less dense than liquid water, these liquids can easily infiltrate and break apart the ice structure. Liquid water molecules have more kinetic energy than ice molecules, so when liquid touches ice, it transfers this energy through collisions, breaking the ice bond. Polar molecules are highly efficient at transferring the heat to the surrounding ice. How to calculate freezing point depression and its application in deicing. -freezing point depression is the reduction of a solvent’s freezing point by adding a non-volatile solute. Formula: Tf= i*Kf*m Term meaning Tf: Freeze point depression, new freezing point for a solvent i: van’t hoff factor. The number of particles a solute breaks into when dissolved. Regular rock salt dissociates into 2 ions so i=2. Kf:(cryoscopic constant):The freezing point depression for water, which is 1.86 celcius * kg/mol, meaning a 1-molal solution of a non-electrolyte lowers the freezing point by 1.86 celsius m: molality of the solution, defined as moles of solute per kilogram of solvent (water) For magnesium chloride, it splits into 3 particles when it dissolves in a solvent, and a higher concentration of it increases the number. For example, a 10 celsius drop in freezing point ( Tf=10) requires approximately 1.99 mol/kg (m) of Mgc1+2 in solution
Variables
Manipulated:
- Types of deicers and components in the deicers
-CMA (Calcium magnesium acetate) (Yardworks Envirosafe™ Ice Melt Jug, Less Harmful To Surfaces, Vegetation, And Pets, 5-kg $11 CAD)
-Calcium chloride (Yardworks High Performance Ice Melter Jug, 5-kg $13 CAD)
-Rock salt/ sodium chloride (Sifto Safe Step Ice Salt Bag, Adds Traction, Rock Salt, 20-kg $8.29 CAD)
-Alcohol water mix ( PSP Isopropyl Alcohol 99% Bottles, 4 x 500 mL $15 CAD, ECOS grape fruit dishsoap $29 CAD (comes in 2 bottles, 3.79 liters and 739ml. Water is too minimal to count it in here since we used tap water)
-Alcohol soap mix ( PSP Isopropyl Alcohol 99% Bottles, 4 x 500 mL $15 CAD, ECOS grape fruit dishsoap $29 CAD (comes in 2 bottles, 3.79 liters and 739ml. Water is too minimal to count it in here since we used tap water)
Responding:
- Time it takes for the ice to melt
- Amount of water left in the pan after the deicer melts it
Controlled:
- Amount of water we freeze for our experiment
- Temperature outside/area we're testing our deicers in
- Type of snow
-9 to -15 celcius range for our experiments. -2 liters of water for pan experiment ( we used titanium and aluminum pots) -1m square and 10cm tall for road experiment -Powder snow
Procedure
Road Experiment Procedure:
1. We first made sure that the temperature was fluctuating between -9 to -15 celcius. This is the temperature we wanted to test our deicers in. It's best not to test it during snowfall but since this was a perfect time for both of us\, we decided to test our deicers during snowfall. We just to put it in a area where snow didn't land on like under a roof or behind a tree. (Snow will accumulate at the top\, which will turn into water. This will affect our results for both the road experiment and steel pan experiment.)
2a. We then prepared the materials that we need for the experiment, this includes : rock salt/sodium chloride, CMA/calcium magnesium acetate, calcium chloride, isopropyl alcohol, warm to hot water, dishsoap, snow shovel, ruler (1m rulers work best), a pencil or a stick, a plastic cup for mixing, a funnel and a weighing scale.

2b. We then prepared our deicers. We measured 50 grams of the dry deicers, which includes CMA/calcium magnesium acetate, calcium chloride and sodium chloride/rock salt. For the alcohol soap mix, we first combined 66ml of isopropyl alcohol, then we added 33ml of warm to hot water and then lastly we added 2 drops of dishsoap. The exact same thing is done with the alcohol water mix, except its a 2:1 ratio of water to alcohol.
Making the alcohol soap mix. (Goes from right to left to down):

Measuring our dry deicers:

3.We then started to make the 5 snow patches that we needed. Since it already snowed for us, we just had to carve a 1m square 5 times through our driveway. We used a pencil to mark the square and a ruler to measure the height, width and length. We made sure the height of the patches were 10cm tall. We then lastly shovelled the excess snow around the snow patches.
 4. For the final step\, we assigned a snow patch a deicer to test. We then we put the deicer beside it for labelling purposes. We then either poured or sprinkled the deicer onto it. We just had to make sure that the deicers were evenly spread across the snow patch.
4. For the final step\, we assigned a snow patch a deicer to test. We then we put the deicer beside it for labelling purposes. We then either poured or sprinkled the deicer onto it. We just had to make sure that the deicers were evenly spread across the snow patch.
Before we applied the deicer:
Top left: CMA
Top right: calcium chloride
Middle right: rock salt/sodium chloride
Bottom left: alcohol soap mix
Bottom right: alcohol water mix

Pan Experiment Procedures: 1.(same): We first made sure that the temperature was fluctuating between -9 to -15 celcius. This is the temperature we wanted to test our deicers in. It's best not to test it during snowfall but since this was a perfect time for both of us, we decided to test our deicers during snowfall. We just to put it in a area where snow didn't land on like under a roof or behind a tree. (Snow will accumulate at the top, which will turn into water. This will affect our results for both the road experiment and steel pan experiment.)
1b.We poured 2 liters of tap water into a metal pan (we highly don't recommend using metal pots and pans but this was simple and easy. We just had to use titanium or aluminum pots or pans because they are more resistant to salt that other kinds of metal.) Then we put the pans outside in our backyard to freeze overnight.

2a. We then prepared the materials that we need for the experiment, this includes : rock salt/sodium chloride, CMA/calcium magnesium acetate, calcium chloride, isopropyl alcohol, warm to hot water, dishsoap, a hanging scale, plastic bag (opitonal), a plastic cup for mixing, a funnel and a weighing scale.

2b.We then prepared our deicers. We measured 10 grams of the dry deicers, which includes CMA/calcium magnesium acetate, calcium chloride and sodium chloride/rock salt. For the alcohol soap mix, we first combined 66ml of isopropyl alcohol, then we added 33ml of warm to hot water and then lastly we added 2 drops of dishsoap. The exact same thing is done with the alcohol water mix, except its a 2:1 ratio of water to alcohol. (DIY deicers will only get one spray so when we test refreeze it won't be the actual deicer were testing for refreeze.)
3.We assigned each pan a deicer for labelling and identification purposes so we knew which one was which similar to the step 4.
Top left: alcohol soap mix
Top right: sodium chloride
Middle: calcium chloride
Bottom left: CMA/calcium magnesium acetate
Bottom right: alcohol water mix

3b. Sprinkle or spray the deicers. (NOTE: our measuring scale isn't in the best shape after the road experiment so the measurements will be a higher than the actual readings. We did keep some of the deicer in the cup or spray bottle to try and balance it out.)

Observations
Road Experiment Observations:
Tested on February 27, from 2:35 pm to 3:55 pm
Our snow patches after 10 minutes at 2:35pm:
Top left: sodium chloride
Top right: CMA
Middle: alcohol water mix
Bottom left: alcohol soap mix
Bottom right: calcium chloride
 The sodium chloride we bought doesn't have any colorants so it blends it with the snow.
The sodium chloride we bought doesn't have any colorants so it blends it with the snow.
Our snow patches after 20 minutes at 2:45pm:
Top left: sodium chloride
Top right: CMA
Middle: alcohol water mix
Bottom left: alcohol soap mix
Bottom right: calcium chloride

Our snow patches after 40 minutes at 3:15pm:
Top left: sodium chloride
Top right: CMA
Middle: alcohol water mix
Bottom left: alcohol soap mix
Bottom right: calcium chloride

Our snow patches after 60 minutes at 3:35pm: Top left: sodium chloride Top right: CMA Middle: alcohol water mix Bottom left: alcohol soap mix Bottom right: calcium chloride

Our snow patches after 80 minutes at 3:55pm: Top left: sodium chloride Top right: CMA Middle: alcohol water mix Bottom left: alcohol soap mix Bottom right: calcium chloride

Comparision of start to finish:
Alcohol soap mix:

CMA:

Calcium chloride:

Rock salt:

Alcohol water mix:

Steel Pan Experiment Observations:
Tested on 12pm on February 28th to 8am on February 29th
Ice in the pan after 60 minutes of deicing at 1pm:
Top left: calcium chloride
Top right: sodium chloride
Middle: alcohol soap mix
Bottom left: CMA
Bottom right: alcohol water mix

Weight of liquid poured from the pans: Top left: calcium chloride =170ml Top right: sodium chloride = 42ml Middle: alcohol soap mix = 188ml Bottom left: CMA =57ml Bottom right: alcohol water mix =39ml

Brine/water after 1 hour at 2pm:
Top left: sodium chloride
Top right: CMA
Middle: alcohol soap mix
Bottom left: alcohol water mix
Bottom right: calcium chloride

Brine/water after 3 hours at 4pm:
Top left: alcohol soap mix
Top right: sodium chloride
Middle: Alcohol water mix (almost fully frozen)
Bottom left: CMA
Bottom right: calcium chloride (starting to freeze)

Brine/water after 7 hours at 8pm:
(It was getting late so we rushed these photos so they ended up blurry)
Top left: alcohol soap mix
Top right: calcium chloride
Middle: alcohol water mix (frozen)
Bottom left: CMA
Bottom right; calcium chloride (almost fully frozen)

Brine/water after 19 hours at 8am:
Top left CMA
Top right: alcohol soap mix
Middle; alcohol water mix
Bottom left: sodium chlordie
Bottom right: calcium chloride
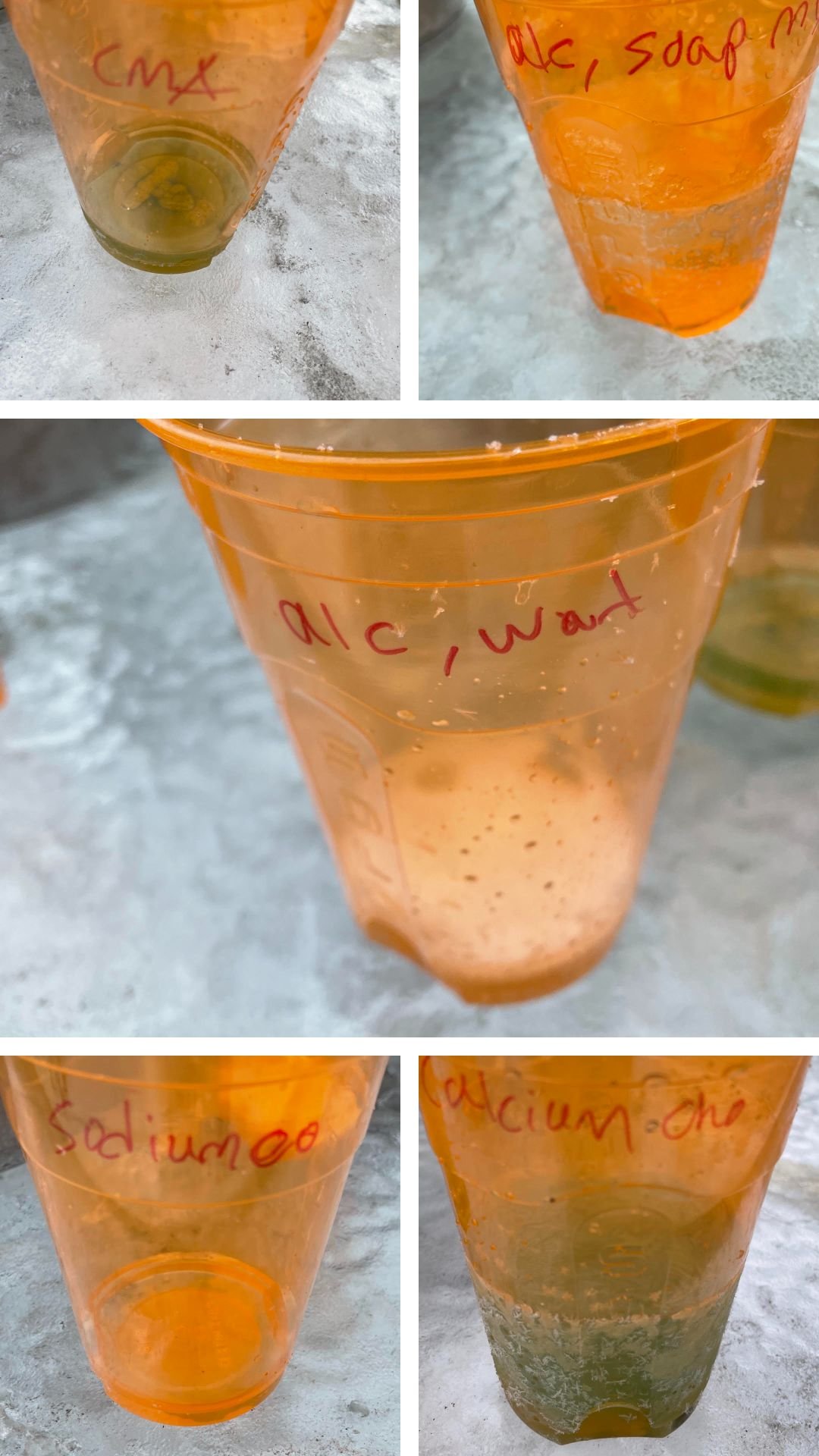
Scores For Road Experiment Test:
These were the scores we decided from the evidence that we had:
Variables for our road experiment:
Manipulated: types of deicers we are testing. (Calcium chloride, rock salt/sodium chloride, CMA/calcium magnesium acetate, alcohol water mix and alcohol soap mix)
Responding: It took around 70 minutes for each deicer to reach its limits.
Controlled: Powder snow, windy and tested around -9 to -11 celcius.
Alcohol soap mix and calcium chloride both scored 1, CMA scored 2, alcohol water mix scored 3 and rock salt/sodium chloride scored last.
We also pulled other factors to determine the winner.
Overall scores in term of speed/efficiency, cost, enviroment impacts and general score:
Speed/efficiency:
| Deicers | Overal Score Out of 10 |
|---|---|
| CMA | 6/10 4th |
| Calcium Chloride | 8.5/10 2nd |
| Alcohol Soap Mix | 9/10 1st |
| Alcohol Water Mix | 7/10 3rd |
| Rock Salt | 2/10 5th |
Cost
| Deicers | Price (per KG or Per Liter in CAD and out of 10) |
|---|---|
| Rock Salt | $0.43 CAD per KG-9/10 1st |
| Calcium Chloride | $2.8 CAD per KG- 5/10 3rd |
| Alcohol Water Mix | \~$6.65 CAD per Liter-3/10 4th |
| Alcohol Soap Mix | \~$6.75 CAD per Liter-3/10 5th |
| CMA | $2.2 CAD Per KG-6/10 2nd |
Notes: isopropyl alchol is not meant to be used in large quantities. Its used for medical or cleaning purposes.
Enviromental Impacts:
| Deicer | How eco-friendly is it?- Out of 10 |
|---|---|
| CMA | 9/10 1st |
| Calcium Chloride | 6.5/10 2nd |
| Rock Salt/Sodium Chloride | 2/10 4th |
| Alcohol Water Mix | 4/10 3rd |
| Alcohol Soap Mix | 1.5/10 5th |
Note: Alcohol water mix scores higher than rock salt and alcohol soap mix because most of its alcohol is diluted with water.
How we calculated the final score:
-5th place = 1 point -4th place = 2 points -3rd place = 3 points -2nd place = 4 points -1st place = 5 points (These include the first line graph results)
Then we add up the points and see which deicer got the most points
CMA: 3,2,4,5 = 14 Calcium chloride: 5,4,3,4 = 16 Rock salt:1,1,5,2 = 9 Alcohol soap mix: 5,5,1,1 = 12 Alcohol water mix:2,3,2,3 = 10
Calcium chloride won this experiment with 16 points.
Scores for pan experiment:
These were the 2 graphs we made using the evidence that we had:
Variables for our pan experiment:
Manipulated: types of deicers we are testing. (Calcium chloride, rock salt/sodium chloride, CMA/calcium magnesium acetate, alcohol water mix and alcohol soap mix)
Responding: It took 5 hours for the alcohol water mix to freeze. It took 16 hours for the calcium chloride brine to refreeze. The alcohol soap mix and sodium chloride were semi-frozen by the end of the 16 hours (some ice formation at the bottom). The CMA was not frozen at all by the end of the 19 hours
Controlled: Powder snow, windy and tested around -10 to -11 celcius, did start warming to -9 at the 20 hour mark. Each brine/water had 20 hours to refreeze
**Note:**10/10 is solid and 0/10 is liquid. (CMA did not show any signs of refreeze in this 16 hours so it stays at the bottom)
CMA is 1st, sodium chloride is 2nd, alcohol soap mix is 3rd, calcium chloride is 4th and alcohol water mix is last in 5th place.
Amount of brine created after the 60 minutes:
(more brine is better which tells us it dissolves ice well. the only exception to this is the DIY deicers, since the deicer itself is liquid. We did test refreeze on the DIY deicers because the deicer itself could freeze. however, the more liquid deicer left, means more dissolving can happen)
| Deicer | Amount of Brine |
|---|---|
| Alcohol water mix | 39ml 5th |
| Alcohol soap mix | 188ml 1st |
| Calcium chloride | 170ml 2nd |
| CMA | 57ml 3rd |
| Sodium chloride | 42ml 4th |
(same) We also pulled other factors to determine the winner.
Overall scores in term of speed/efficiency, cost, enviroment impacts and general score:
Speed/efficiency:
| Deicers | Overal Score Out of 10 |
|---|---|
| CMA | 6/10 4th |
| Calcium Chloride | 8.5/10 2nd |
| Alcohol Soap Mix | 9/10 1st |
| Alcohol Water Mix | 7/10 3rd |
| Rock Salt | 2/10 5th |
Cost
| Deicers | Price (per KG or Per Liter in CAD and out of 10) |
|---|---|
| Rock Salt | $0.43 CAD per KG-9/10 1st |
| Calcium Chloride | $2.8 CAD per KG- 5/10 3rd |
| Alcohol Water Mix | \~$6.65 CAD per Liter-3/10 4th |
| Alcohol Soap Mix | \~$6.75 CAD per Liter-3/10 5th |
| CMA | $2.2 CAD Per KG-6/10 2nd |
Notes: isopropyl alchol is not meant to be used in large quantities. Its used for medical or cleaning purposes.
Enviromental Impacts:
| Deicer | How eco-friendly is it?- Out of 10 |
|---|---|
| CMA | 9/10 1st |
| Calcium Chloride | 6.5/10 2nd |
| Rock Salt/Sodium Chloride | 2/10 4th |
| Alcohol Water Mix | 4/10 3rd |
| Alcohol Soap Mix | 1.5/10 5th |
Note: Alcohol water mix scores higher than rock salt and alcohol soap mix because most of its alcohol is diluted with water.
**we used the same calculation method**
CMA: 5,3,2,4,5 = 19 Calcium chloride: 2,4,3,4,4 = 17 Rock salt:4,1,5,2,2 = 14 Alcohol soap mix: 3,5,1,1,5 = 15 Alcohol water mix:1,3,2,3,1 = 10
CMA is the winner for the pan experiment
Total scores added up from both experiments
| 1st CMA and calcium chloride | 33 points |
|---|---|
| 2nd alcohol soap mix | 27 points |
| 3rd sodium chloride | 23 points |
| 4th alcohol water mix | 20 points |
Analysis
Road Experiment Analysis:
Rock salt/sodium chloride: It didn't do well for many reasons. It first had a lack of colorants, which made it very hard for us to rate it because we can't touch the experiments. It also was technically colder than the recommended temperature which slowed the dissolving speed. Another reason is because its too dry to start creating a brine. Since it was very dry that day, many of the dry deicers took a while to start the brining process. However, since the rock salt were made in large clumps, the kinetic energy it had when falling into the snow was enough to go deep into the snow. Rock salt becomes ineffective in deep snow.
Calcium chloride: Calcium chloride is a powerhouse at melting both ice and snow. It was able to do well in this experiment. The vibrant blue colours it had made it easy for us to rate it. Its initial brining process is also extremely fast, even with the dry conditions. It also generates heat which also speeds up the dissolving rate.
CMA/calcium magnesium acetate: CMA did not do good but also did not do bad. It's in the middle of all the deicers you could choose. Its recommended temperature is not extremely low but it is eco-friendly and doesn't corrode metal as fast as chlorides. Its results were expected.
Alcohol water mix Alcohol water mix started off good but slowed a lot by the end. Its intial heat it had was enough to melt away the top layer of snow but the instant cooling of the water benefited ice formation rather than ice melting. The isopropyl alcohol ratio was not enough and could not melt the ice further. A fix to this could be using hotter water or more isopropyl alcohol
Alcohol soap mix: This deicer did very well. It had same issues with water cooling with the alcohol water mix but the larger ratio of isopropyl alcohol managed to continue dissolving. Most of the deicer ended up in the same holes because we poured the liquid deicer instead of spraying it, this led most of the deicer into the concrete rather in the snow.
Pan Experiment Analysis:
Rock salt: It did decently at ice melting which we expected. The results with the refreeze was not expected. The sodium chloride managed to stay liquid longer than calcium chloride. This is probably the low amounts of brine it created only touched a small amount of plastic surface area which cools slower. Another reason for this is because calcium chloride already has slush poured into it which lowered its temeperature.
Calcium chloride: It did well at melting the ice. We even had a spillage issue when some of the brine leaked out of the cup when we poured it and it was able to create more brine that the other dry deicer. It did freeze faster which was because of a larger surface area which means it cools faster.
CMA: Both the ice melting and refreeze were expected. It was able to break apart the top layer of ice and create divots in the ice. CMA is able to keep brine in a liquid form for many hours.
Alcohol water mix: The intial hot water was able to melt through the top layer, however, it instantly cooled and the small amounts of isopropyl alcohol was barely able to keep it liquid. This is why we ended up with slush in our cup rather than liquid. This means it was intially cooler than the other deicers, this is why it freezed first.
Alcohol soap mix: The water and isopropyl alcohol melted around the pan and cracked the ice into many segments. Just like the water cooling issue, the hot water instantly turned to freezing cold water, however, a higher ratio of isopropyl alcohol was able to keep it liquid. This ended up leaving the isopropyl alcohol to sit there without dissolving anything. Most of the alcohol went into the cup we used to test refreeze, this is why it was able to stay liquid for so long.
Conclusion
General Conclusion:
We concluded that our general hypothesis was half correct. The Calcium Chloride is the best deicer to use in typical Calgary weather, however, CMA also scored first meaning we have 2 winners. Calcium chloride is adaptable and usable in various temperatures. It melts efficiently and quickly, and can melt and function properly in ice and snow. Though it did refreeze after a bit of time due to the bigger pan that the ice was placed on, we can now predict that with the results that it has shown, it would have refroze much slower than before. This deicer can be used anywhere from 0°C to (-32)°C and is recommended for deep snow and ice. CMA is enviromentally friendly and non corrosive. It average in melting ice and snow but it is used without concern of damaging concrete, vegetation and cars. To make our road experiments more accurate next time, we could have used a meter box to indent our lines for a more accurate snow patches and more exact measurements. To make our pan experiment more accurate next time, we could use that exact same brand, style and material of pans and have a better area to do our experiments. (We tested this on a small table to prevent our dogs from ingesting any sort of toxic deicer. This however made the area quite tedious to move around which led us to spill some deicer.)
Chloride, Acetate and Homemade deicers Conclusion:
Our chloride hypothesis was correct. Calcium chloride is adaptable and usable in various temperatures. It melts efficiently and quickly, and can melt and function properly in ice and snow. Though it did refreeze after a bit of time due to the bigger pan that the ice was placed on, we can now predict that with the results that it has shown, it would have refroze much slower than before. This deicer can be used anywhere from 0°C to (-32)°C and is recommended for deep snow and ice. rock salt is, however very cheap but its not recommended for low temperature use.
Our acetate was correct because CMA was the only acetate we tested. (sodium acetate and potassium acetate can be bought straight from the factory, however, the deicer alone is extremely expensive plus the delivery fee adds up to more than 100 CAD).
Our DIY deicer hypothesis is correct. The alcohol soap mix had more isopropyl alcohol, the main deicing agent. The alcohol water mix had too much water making it ineffective at low temperatures.
Extra conclusions for each deicer:
For the Rock Salt/Sodium Chloride, when we did our experiment, the temperature was lower than the recommended one, so it slowed down the melting process. It also lacked colorants, which made it harder for us to rate. It is also inefficient in deep snow or dry areas. It is more efficient in ice, however, as it does have a slower re-freezing time/rate. This deicer is useful anywhere from 0°C to (-7)°C and in more dense places, and specifically more for ice than snow. Only con, it is not eco-friendly and highly corrosive.
As for the Calcium Magnesium Acetate, it is a deicer you would use for any "mid" temperatures. As you can see with the results, it didn't do horrible but it didn't do great. It is essentially the "medium" or "in-between" deicer people would use. Its recommended temperature isn't really low, but is a great eco-friendly, non-corrosive option. It is okay for ice, but not deep snow and does not refreeze too quickly. Its recommended temperature is anywhere from (-7)°C or higher.
The alcohol and water mix probably did the worst out of all the other deicing agents. This one started off strong, but as it melted the top layer of ice, the coolness of the water got to it and actually started to form ice and not melt it. You could use hotter water or more alcohol to help things a little more. Because of this, it ended up in a slush and unsurprisingly was the first to refreeze. We tested this out because this hack was going viral on the internet and a lot of people were successful with it. We do not recommend it as it is cheap but doesn't help melt the ice and does the complete opposite of its initial function and isn't eco-friendly either.
The Alcohol Soap mix did surprisingly well, considering that this deicer and the one above are both alcohol based. At first it had some problems with water cooling, but an extra addition of alcohol made it dissolve better. It also didn't refreeze as quickly as well. We recommend using this in any temperature from (-18)°C or lower, and is goof for thin ice or thin/small amounts of snow. The only con to this is that it is not eco-friendly.
Extra points/notes
These scores cannot determine if it is better or not. Someone might value money over efficiency or someone might value a non corrosive deicer. Because we thought calcium chloride was the best doesn't mean it applies for everyone. Some people will just stick to rock salt for its price and some people will only stick to DIY deicers. This project was for us to give people an idea of deicers, plus an general idea of the best deicer.
Application
The information we gathered through our project can be used during Calgary winter times right after it snows, after it has rained or the snow has melted and refroze, or when there's thick ice. When people are having trouble deciding which deicer to use and which would be efficient, they can refer back to our findings. People can use it for their driveways or sidewalks, in bigger cases on places like the airport runway or bridges and such.
Sources Of Error
General sources of error in our experiments:
- Our CMA (calcium magnesium acetate) deicer that we chose has different types of chlorides and chemicals. This alters our experiments for CMA because it contains urea, rock salt and calcium chloride. This goes for every other dry deicers. (but if for example I was testing calcium chloride, it would be mostly calcium chloride with some other chemicals. It wouldn't be a mix of everything.)
- Temperatures ussualy fluctuate constantly, meaning each result will have a bit of altered results.
- Wind and moisutre affects our experiments by either accelerating or decelerating refreeze and how well our deicers acutally work.
- Rating scores out of 10 is unreliable and is not the most accurate tool to measure how well that deicer did
- How we measured the scores is not the most reliable method and there are definetly better methods.
Steel pan experiments sources of error:
- Each cup has a different amount of brine or deicer, meaning less liquid equals to less surface area meaning its harder to freeze than large amounts of liquid. (This is why the rock salt brine was able to stay in liquid form longer than calcium chloride brine.)
- We did have some slush poured into the calcium chloride cup which dissolved before we could get it out, this led the intial starting temperature to be colder than others.
- The measuring scale did start to malfunction after the road experiment so we ended with more deicer than what we acutally wanted. We left some deicer in the cup or spray bottle to balance it out.
Road experiment sources of error:
- During the CMA and the 2 DIY deicer test, there was more wind then for the other experiments. The DIY deicers lost alot of its amount due to winds during spraying.
- Each snow patch might not be 10cm throughout making the results unfair. Each snow patch is also different in size.
- The wind blew snow into our square patches of snow which then accumulated at the top, this extra snow made it harder for us to judge it.
Citations
- Neighbourhood Heroes. (2026, January 31). What are eco-friendly alternatives to salt for snow melting?. Neighbourhood Heroes Lawn Care. https://web.neighbourhoodheroes.ca/blog/eco-friendly-snow-melting-alternatives
- Kimbrough, D. (2006). Salting Roads. https://passchemistry.com/wp-content/uploads/2020/02/chemmatters-feb2006-salting-roads.pdf
- Roads. (n.d.). Road salt management plan. Https://Www.calgary.ca. https://www.calgary.ca/roads/conditions/snow-ice-salt.html
- Exploring the Dangers of Road Salt & Dog Paws. (2025, November 5). Vetericyn Animal Wellness. https://vetericyn.com/blogs/vetericyn/salt-and-dogs-paws
- discountsnowstakes. (2024, May 31). 10 Cheapest Ways to Salt a Property: Effective and Budget-Friendly Solutions - Driveway Markers News. Driveway Markers News. https://discountsnowstakes.com/blog/10-cheapest-ways-to-salt-a-property-effective-and-budget-friendly-solutions/?srsltid=AfmBOooh6oA5Xs3hN2HqcbFmvMr-_8UWaVV64c2R8DDP1y8KesHbTxwk
- FSI. (2021, February 7). All about ice melt - FSI Landscape Supply. FSI Landscape Supply. https://fsilandscapesupply.com/salt/all-about-ice-melt/
- Loeffler, B. (2022, December 20). The Chemistry of Deicing: How Does Ice Melt Work? Redmond.com; Redmond Life. https://blog.iceslicer.redmond.com/the-chemical-composition-of-deicers
- Rollin, J. (2024, January 14). Deicers Efficiency Overview: What Melts Ice the Fastest? Ninja De-Icer. https://ninjadeicer.com/blogs/resources/de-icers-efficiency-overview-what-melts-ice-the-fastest
- Is An Asphalt Driveway Cheaper Than Concrete? | EMESA. (2024\, October 21). Emesa | Asphalt Paving & Sealant | Concrete Sealant \, Driveways & Stamping Concrete. https://emesa.ca/is-an-asphalt-driveway-cheaper-than-concrete
- Hick's Paving. (2024, November 27). How Asphalt Driveways Beat Concrete in Snow Melt Efficiency. Hicks Paving. https://www.hickspavinginc.com/post/how-asphalt-driveways-beat-concrete-in-snow-melt-efficiency
- EPA. (2024, December 16). Winter is Coming! And with it, tons of salt on our roads. Www.epa.gov. https://www.epa.gov/snep/winter-coming-and-it-tons-salt-our-roads
- Does higher humidity cause ice to melt faster? (2019). Quora. https://www.quora.com/Does-higher-humidity-cause-ice-to-melt-faster-1
- Anti-icers and Deicers (chemicals) Selection Guide: Types\, Features\, Applications | GlobalSpec. (2026). Globalspec.com. https://www.globalspec.com/learnmore/manufacturing_process_equipment/cleaning_surface_preparation/antiicers_deicers_chemicals
- Liquid Deicing vs Rock Salt | Levan Machine and Truck Equipment. (2023, September 26). Levan Machine and Truck Equipment. https://levanmachine.com/snow-plows/liquid-deicing-vs-salt/
- Loeffler, B. (2023, July 7). How Do Deicers Affect Biochemical Oxygen Demand? Redmond.com; Redmond Life. https://blog.iceslicer.redmond.com/how-do-deicers-affect-biochemical-oxygen-demand
- Corrosion inhibitor. (2020, February 11). Wikipedia. https://en.wikipedia.org/wiki/Corrosion_inhibitor
- https://westerntransportationinstitute.org/wp-content/uploads/2018/02/4W4758_Non-choride-deicers_-Report-Brief.pdf
- Scientists Use Agricultural Waste to Develop New Road Deicer. (n.d.). Yale E360. https://e360.yale.edu/digest/scientists-use-agricultural-waste-to-develop-new-road-deicer
- Green Purchasing Best Practices: Deicers Prepared for: Washington State Department of Enterprise Services (DES) National Association of State Procurement Officials (NASPO) Research and Writing by: Responsible Purchasing Network. (2013). https://www.responsiblepurchasing.org/purchasing_guides/deicers/naspo_rpn_deicers_purchasing_guide.pdf
- Gilmore, R., & Kirby, D. (2025, June 6). The best car de-icer for clearing frost and ice quickly and effectively. Parkers. https://www.parkers.co.uk/car-advice/car-care/best-de-icer/
- Blog | Imperial Dade. (n.d.). Www.imperialdade.com. https://www.imperialdade.com/blog/liquid-ice-melt
- Schneider, J. (2025, October 27). Types of Ice Melt: Benefits and Drawbacks Explained. Brody Chemical. https://brodychemical.com/blogs/guides/types-of-ice-melt
- Tips to use ice melt products properly and lower the risks they pose to pets, property. (2018). Accuweather.com. https://www.accuweather.com/en/weather-news/tips-to-use-ice-melt-products-properly-and-lower-the-risks-they-pose-to-pets-property-2/433875
- Partin, M. (2024). Applying The Right Amount Of De-Icer Will Save You Money & Protect Our Water From Contamination. Whygoodnature.com. https://www.whygoodnature.com/blog/applying-the-right-amount-of-de-icer-will-save-you-money-protect-our-water-from-contamination
- The 4 Best Ice Melts. (n.d.). GearLab. https://www.techgearlab.com/topics/home/best-ice-melt
- Process Applications Calcium Chloride Brine Series Overview Calcium Chloride Series Brines Calcium chloride-based heat transfer fluids Dynalene’s Fluid Care Program. (n.d.). https://www.dynalene.com/wp-content/uploads/2018/07/Dynalene_Calcium_Chloride_Series_Technical_Data_Sheet.pdf
- Wikipedia Contributors. (2019, December 2). Isopropyl alcohol. Wikipedia; Wikimedia Foundation. https://en.wikipedia.org/wiki/Isopropyl_alcohol
- (2025). Istockphoto.com. https://www.istockphoto.com/photos/liquid-deicer
- Johnston, B. (2022, September 29). How to Use Ice Melt on Sidewalks. Tomahawk Power. https://tomahawk-power.com/blogs/articles/how-to-use-ice-melt-on-sidewalks?srsltid=AfmBOoorpSPHawv47vUIMoKu4isoo_nsk4OgIaldeXZfB5PKl9EUzhpb
Acknowledgement
Firstly, we'd like to thank our CYSF coordinators, Mr. Lahoda, Ms. Allen and Mr. Winter.
Next we'd like to thank our parents for aiding in materials for our experiments and giving us tips that helped us create this project. They put in money to buy our materials and deicers for our experiment to work.
We'd also like to thank Aryan, who helped give us ideas and project tips that helped us improved the overall quality of our project.
Finally, we'd thank all the people who wrote the research papers that we used in our project.

