Are There Really "Butterflies" in Your Stomach? A Systematic Review of the Neurobiology of Romantic Love
Abdul Shahzad
Sir Winston Churchill High School
Grade 10
Presentation
No video provided
Problem
Research Question
Which brain regions show the most consistent activations during romantic love in existing fMRI studies?
Signifigance
Understanding the neural processes involved in romantic love helps bridge the psychological idea of “love” to biological mechanisms. Identifying consistent patterns of brain activation can contribute to a clearer framework for romantic attachment and may inform related fields such as therapy, mental health, and the study of affective disorders. This review aims to summarize existing fMRI and neuroscience research to identify the brain structures most involved in romantic love and connect these various studies to construct a more comprehensive model of its neural basis
Hypothesis
If romantic love is present in a subject, then it will be associated with activations in regions involved in reward and motivation, such as the dopaminergic reward pathway, because romantic love functions as a goal-directed motivational state characterized by sustained attention and attentional bias towards a specific individual. Therefore, higher blood oxygen level-dependent (BOLD) signals are expected in neural circuits underlying reward and motivational processing.
Method
Paper Acquisition
Papers used in this review were acquired using PubMed, a publicly accessible database that includes most, if not all, published, reviewed research papers and their citations. Using PubMed, the following search query was used: ("romantic love"[Title/Abstract] OR "love"[Title/Abstract]) AND ("fmri"[Title/Abstract]). This returned 73 results. Afterwards, the papers were screened with the predefined inclusion and exclusion criteria (see Inclusion and Exclusion Criteria). This left nine citations that were originally found in PubMed. Two more papers that followed the criteria were found from source lists of cited papers included in this review; the total number of papers used in this review was eleven. The papers were searched for on Google Scholar and then downloaded as PDFs for analysis and future reference.
Inclusion and Exclusion Criteria
All papers included in this review met predefined criteria to ensure quality and accuracy. Papers were first included if they were peer-reviewed, focused on romantic love using fMRI, available in English, published between 2000 and 2025, and reported neurological findings. Papers were excluded if they reported findings unrelated to romantic love, such as platonic or familial love, studies that did not use fMRI, the full text was not freely accessible, studies using animal subjects, and reviews, designs, or meta-analyses. The paper selection process followed principles consistent with PRISMA guidelines.
Study Selection Process
Using the above criteria, papers were filtered with different methods. Firstly, the PubMed checkbox selection menu was employed to first filter the dates of publication, type of paper, and if full-text access was available for free. After this, 17 papers remained. During the process of analyzing the papers, the main focus of the paper, identified from the abstract, was then used to further narrow down the total number of papers to nine. Papers that matched the question of this research project that were found from the citations of the initial papers were also included if they passed all inclusion and exclusion criteria. Neural activation reported in this review refers to increased blood-oxygen-level-dependent (BOLD) signal measured through functional MRI, as defined by the original studies.
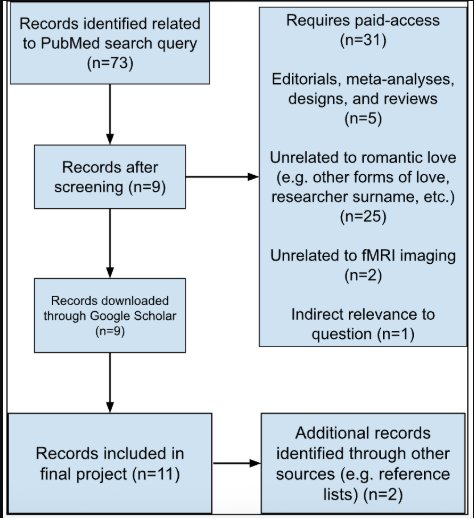 Figure 1. PRISMA flow diagram summarizing identification, screening,
and exclusion of papers in this review.
Figure 1. PRISMA flow diagram summarizing identification, screening,
and exclusion of papers in this review.
Research
Romantic love as a neural phenomenon
Romantic love is often seen as an emotion, similar to happiness, anger, or sadness. However, neuroscience suggests that romantic love is actually a motivational drive involving reward-processing regions such as the ventral tegmental area (VTA) and caudate nucleus (Aron et al., 2005; Fisher et al., 2005). Romantic love involves focused attention and goal-directed behavior towards a specific individual that a person loves (Langeslag et al., 2014). Because of these unique characteristics, researchers continue to hypothesize about the connection between romantic love and reward processing and motivation pathways.
Use of fMRI in studying romantic love
Functional magnetic resonance imaging (known as fMRI) measures live brain activity using blood oxygen level dependent (BOLD) signals, generated by the consumption of oxygen and influx of oxygen-rich blood to neurons when active, indicating neuronal function. Researchers have different means of eliciting this reaction and simulating the feeling of romantic love, but the most common practice is using images of the participant’s beloved (provided by them) and a control, such as an image of a friend similar to the beloved or a stranger, to compare the activations. This comparison allows researchers to isolate neural activity specifically associated with romantic attachment while controlling for visual and social processing regions unrelated to romantic emotion, such as regions for facial recognition or vision.
Previously reported brain regions
Previous studies have reported activations during romantic love in regions that are associated with reward and motivation pathways, as hypothesized. For example, regions in the midbrain, such as the ventral tegmental area (VTA) (Acevedo et al., 2020; Aron et al., 2005), as well as components of the basal ganglia (Eckstein et al., 2023). However, regions involved in emotional processing have also been observed to produce BOLD signals, such as the insular cortex (Acevedo et al., 2012; Bartels & Zeki, 2004; Ebisch et al., 2014) and the anterior cingulate cortex (ACC) (Acevedo et al., 2012; Bartels & Zeki, 2004; Eckstein et al., 2023). However, reported findings vary across studies.
Gap in the literature
Although many studies examine romantic love, there is variability in activation patterns and results reported due to many variables, such as sample size or experiment methodology. Few analyses systematically compare which regions appear most consistently across studies to identify convergent BOLD signals. Therefore, a structured literature review is necessary to identify overlapping neural activation patterns in order to better understand the underlying connection of romantic love and the brain.
Data
Results
Brain Regions
Across the analyzed papers, several regions were consistently reported as activated or deactivated during romantic love. For example, the insular cortex (also known as the insula) was found to be activated in six out of eleven studies analyzed. The anterior cingulate cortex, in proximity to the insula, was also observed to have an activation signal during romantic love in four studies. Also analyzed as activating during romantic love was the dorsal striatum, the area including the caudate nucleus and putamen, which had reported activation five times and four times respectively across eleven papers, particularly in those that had asked participants to view images of the person they love romantically versus a control. Another region that was discovered to activate during romantic love was the ventral tegmental area (VTA), activating in four out of eleven studies gathered. Lastly, there is the amygdala, which was seen to both activate and deactivate in certain contexts of romantic love, depending on the experiment, reported in five studies out of the total eleven. Other regions of the brain also reported BOLD signals (thalamus, etc.); however, they were not as consistent across the eleven studies as the aforementioned regions were. Most regions that reported activation in the studies analyzed are included in the figure or table below.

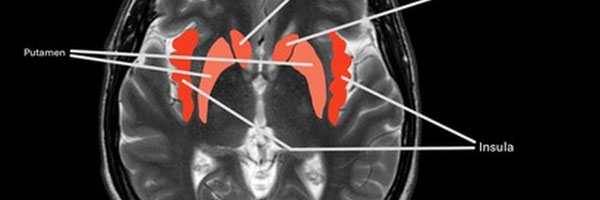
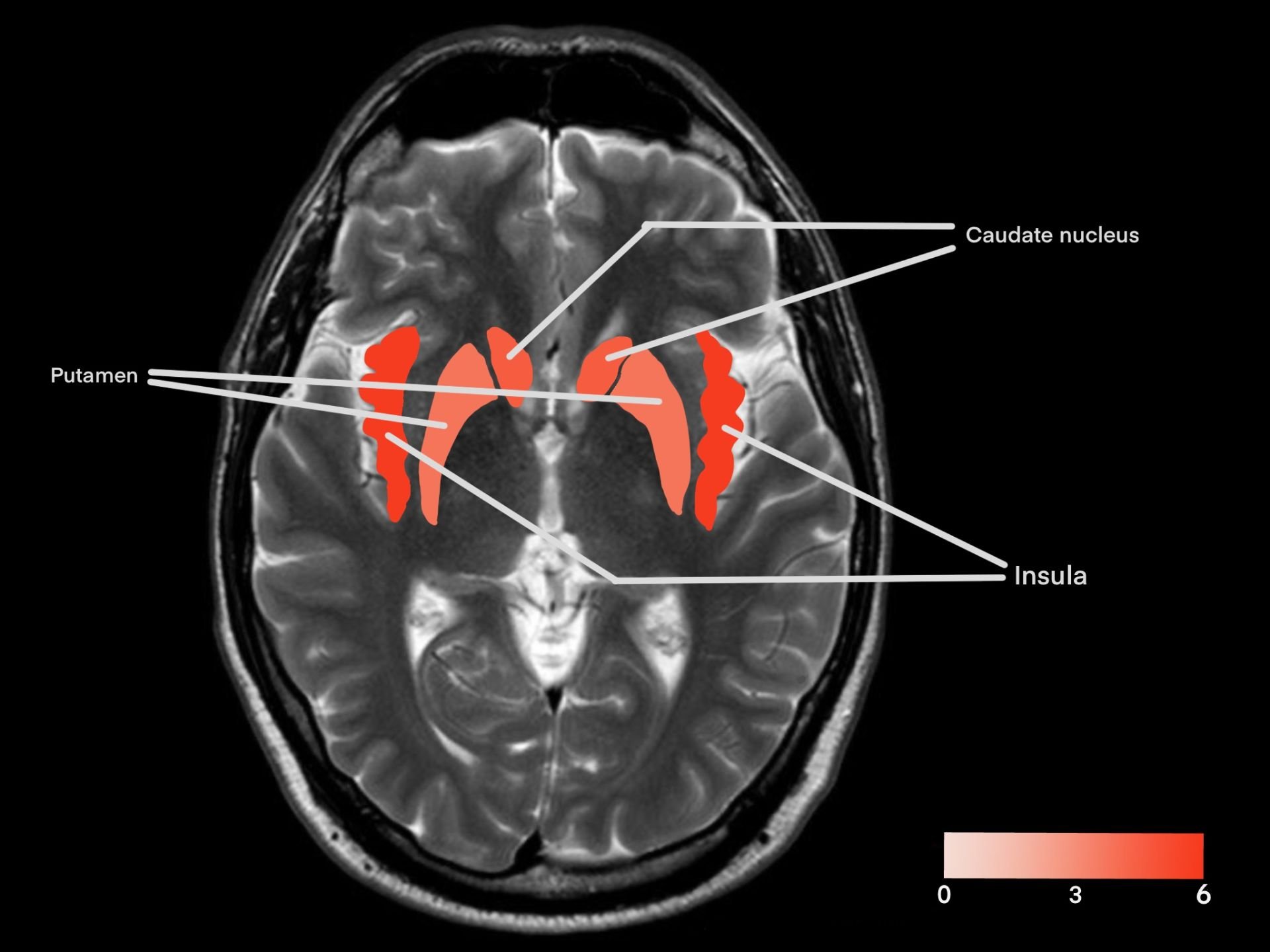 Figure 2A/2B. Frequency of reported activation across 11 fMRI studies examining romantic love. Color intensity represents the number of studies reporting activation in each region.
Figure 2A/2B. Frequency of reported activation across 11 fMRI studies examining romantic love. Color intensity represents the number of studies reporting activation in each region.
| Brain region | Number of BOLD activations reported in studies (n=11) |
|---|---|
| Insular cortex (insula) | 6 |
| Caudate nucleus (caudate) | 5 |
| Ventral tegmental area (VTA) | 4 |
| Putamen | 4 |
| Anterior cingulate cortex (ACC) | 4 |
| Cerebellum | 3 |
| Thalamus | 3 |
Table 1. Frequency of reported BOLD activations reported across 11 fMRI studies of romantic love. Values represent the number of studies identifying significant activation in each region. Regions that did not appear more than twice were not included.
Conclusion
Discussion
Across the fMRI studies analyzed, the majority of consistently activated regions have been associated with limbic, reward, or motivation pathways in the brain. These findings support the hypothesis that romantic love involves long-term attention, goal-directed behaviour, and attentional bias, engaging neural systems underlying these processes.
Motivational and reward-related brain systems in romantic love
A region that consistently showed activation across studies was the ventral tegmental area (VTA), an area of the brain that is involved in motivation and reward processing (Fitzgerald & Day, 2024). VTA activation was observed across studies when participants viewed images of their loved ones or engaged in positive reflection (Acevedo et al., 2012, 2020; Aron et al., 2005; Poore et al., 2012), such as in Poore et al. (2012) where participants were asked to imagine what compliment their partner would give them, which resulted in increased VTA activity. These findings are consistent with the role the VTA plays in reward pathways and motivation, suggesting that romantic love engages neural systems underlying reward and motivation.
Within the same reward pathway as the VTA is the caudate nucleus, which is involved in goal-directed reward learning and positive reinforcement (Haruno et al., 2004), thereby reinforcing behaviours associated with reward outcomes. The caudate nucleus was observed to activate in response to exposure to the participant’s beloved, whether anticipating viewing an image of them, actually viewing their image, or thinking of experiences that they shared (Acevedo et al., 2012; Aron et al., 2005; Bartels & Zeki, 2004; Langeslag et al., 2014). For example, in Langeslag et al. (2014), the caudate nucleus in participants had a significantly greater activation signal when anticipating seeing an image of their beloved compared to waiting for an image of their friend or a stranger. Within the same circuit as the caudate (together being the dorsal striatum), the putamen has been associated with stimulus-response learning and habit reinforcement (Ersche et al., 2021). Studies examining romantic love also reported putamen activation, such as in the previously mentioned experiment in Langeslag et al., during anticipation of receiving a compliment. Together, activation of the caudate nucleus and putamen suggests that romantic love engages dorsal striatal circuitry involved in both goal-directed behaviour and habit reinforcement, potentially supporting the persistence of attachment-like behaviours over time.
Emotional processing systems in romantic love
Alongside the reward and motivational processing of romantic love, there is also an emotional aspect involved, supported by the results of the analyzed studies.
A region of the brain that is often associated with emotional response is the insular cortex (also known as the insula), which is involved in processing internal functions like heartbeat and breathing, integrating bodily states into emotions like fear, disgust, or, in this case, romantic love (Uddin et al., 2017; Zhang et al., 2024). The insula was the most frequently activated region across the analyzed studies, particularly in experiments where participants were asked to report their personal feelings after the experiment (Acevedo et al., 2012; Bartels & Zeki, 2004; Rinne et al., 2024). For example, in Ebisch et al. (2014), participants were either caressed with a rubber ball (for control) or the hand of their beloved. These findings indicate that insular activity was increased during the trial using the hand but not the ball. This suggests that the physical caress associated with their beloved contributed to the increased activation, supporting the current consensus on its function with bodily monitoring and emotion. This suggests that romantic love is not simply abstract cognition, but also involves bodily states.
Another region implicated in emotional processing is the anterior cingulate cortex (ACC), which is involved in not only processing but also emotional regulation, attentional control, and affective processing (LaPrarie et al., 2011). Studies in which the insula was activated frequently also reported activation in the ACC. This supports the current idea that the insula and ACC work in tandem to process emotions (Medford & Critchley, 2010), supporting the interpretation that both regions play a coordinated role in the emotional processing of romantic love.
Deactivations during romantic love
Although many regions show increased activation, some regions demonstrate decreased BOLD activity during romantic love and are important in understanding the neural mechanisms of romantic love. The amygdala was frequently reported as deactivated across studies examining romantic love (Aron et al., 2005; Bartels & Zeki, 2004). However, some experiments observed increased amygdala activity under specific conditions. The amygdala is a region of the brain associated with aggression, anxiety regulation, threat detection, and social cognition (Qais et al., 2023). Decreased amygdala activity in a subject may reflect reduced social vigilance or threat monitoring toward their romantic partner, which may facilitate attachment and trust. In contrast, increased amygdala activity in certain examples, such as during the anticipation of partner feedback (Eckstein et al., 2023), may indicate heightened emotional arousal or salience rather than fear-related processes. This pattern of activation and deactivation suggests that romantic love does not entirely subdue amygdala activity, but rather is context-dependent.
Limitations
The number of analyzed studies in this review was relatively small, at eleven total included. The studies analyzed also had different sample sizes, demographics, relationship lengths, and other variables. These differences may influence the summarized activation patterns. The studies also used different control conditions and imaging parameters that may contribute to variability in reported activation.
BOLD signals that were used to identify activations reflect indirect measures of neural activity and do not provide information about neurotransmitter release or causal mechanisms. BOLD signals do not reflect direct neural activation, but instead the oxygen consumed during imaging, and it is inferred that the region would then be active.
Studies reporting significant activation patterns may be more likely to be published, potentially biasing the available literature.
Future applications
Understanding consistent neural activation patterns associated with romantic love may help inform clinical approaches to attachment-related disorders and mood disorders involving reward-processing dysfunction, possibly creating a better foundation for treatment and care. Identifying similar activation patterns may also guide future neuroimaging studies toward more standardized methodologies and targeted investigation of specific neural regions. It also has broader neuroscience implications because romantic love represents a complex social state and can serve as a model for studying human attachment, social bonding mechanisms, and motivation in social contexts.
Citations
Citations
Acevedo, B. P., Aron, A., Fisher, H. E., & Brown, L. L. (2011). Neural correlates of long-term intense romantic love. Social Cognitive and Affective Neuroscience, 7(2), 145–159. https://doi.org/10.1093/scan/nsq092
Acevedo, B. P., Poulin, M. J., Collins, N. L., & Brown, L. L. (2020). After the Honeymoon: Neural and Genetic Correlates of Romantic Love in Newlywed Marriages. Frontiers in Psychology, 11, 493805. https://doi.org/10.3389/fpsyg.2020.00634
Aron, A., Fisher, H., Mashek, D. J., Strong, G., Li, H., & Brown, L. L. (2005). Reward, Motivation, and Emotion Systems Associated With Early-Stage Intense Romantic Love. Journal of Neurophysiology, 94(1), 327–337. https://doi.org/10.1152/jn.00838.2004
Bartels, A., & Zeki, S. (2004). The neural correlates of maternal and romantic love. NeuroImage, 21(3), 1155–1166. https://doi.org/10.1016/j.neuroimage.2003.11.003
Ebisch, S. J., Ferri, F., & Gallese, V. (2014). Touching moments: Desire modulates the neural anticipation of active romantic caress. Frontiers in Behavioral Neuroscience, 8, 67592. https://doi.org/10.3389/fnbeh.2014.00060
Eckstein, M., Stößel, G., Gerchen, M. F., Bilek, E., Kirsch, P., & Ditzen, B. (2023). Neural responses to instructed positive couple interaction: An fMRI study on compliment sharing. Social Cognitive and Affective Neuroscience, 18(1). https://doi.org/10.1093/scan/nsad005
Ersche, K. D., Lim, T. V., Murley, A. G., Rua, C., Vaghi, M. M., White, T. L., Williams, G. B., & Robbins, T. W. (2021). Reduced Glutamate Turnover in the Putamen Is Linked With Automatic Habits in Human Cocaine Addiction. Biological Psychiatry, 89(10), 970–979. https://doi.org/10.1016/j.biopsych.2020.12.009
Fisher, H., Aron, A., & Brown, L. L. (2005). Romantic love: An fMRI study of a neural mechanism for mate choice. Journal of Comparative Neurology, 493(1), 58–62. https://doi.org/10.1002/cne.20772
Fitzgerald, N. D., & Day, J. J. (2025). Neuronal heterogeneity in the ventral tegmental area: Distinct contributions to reward circuitry and motivated behavior. Addiction Neuroscience, 14, 100191. https://doi.org/10.1016/j.addicn.2024.100191
Haruno, M., Kuroda, T., Doya, K., Toyama, K., Kimura, M., Samejima, K., Imamizu, H., & Kawato, M. (2004). A Neural Correlate of Reward-Based Behavioral Learning in Caudate Nucleus: A Functional Magnetic Resonance Imaging Study of a Stochastic Decision Task. The Journal of Neuroscience, 24(7), 1660–1665. https://doi.org/10.1523/jneurosci.3417-03.2004
Langeslag, S. J. E., van der Veen, F. M., & Röder, C. H. (2012). Attention modulates the dorsal striatum response to love stimuli. Human Brain Mapping, 35(2), 503–512. https://doi.org/10.1002/hbm.22197
LaPrairie, J. L., Schechter, J. C., Robinson, B. A., & Brennan, P. A. (2011). Perinatal risk factors in the development of aggression and violence. Advances in Genetics, 75, 215–253. https://doi.org/10.1016/b978-0-12-380858-5.00004-6
Medford, N., & Critchley, H. D. (2010). Conjoint activity of anterior insular and anterior cingulate cortex: awareness and response. Brain Structure & Function, 214(5), 535. https://doi.org/10.1007/s00429-010-0265-x
Poore, J. C., Pfeifer, J. H., Berkman, E. T., Inagaki, T. K., Welborn, B. L., & Lieberman, M. D. (2012). Prediction-error in the context of real social relationships modulates reward system activity. Frontiers in Human Neuroscience, 6. https://doi.org/10.3389/fnhum.2012.00218
Qais AbuHasan, Reddy, V., & Siddiqui, W. (2023, July 17). Neuroanatomy, Amygdala. Nih.gov; StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK537102/
Rinne, P., Lahnakoski, J. M., Saarimäki, H., Tavast, M., Sams, M., & Henriksson, L. (2024). Six types of loves differentially recruit reward and social cognition brain areas. Cerebral cortex (New York, N.Y. : 1991), 34(8), bhae331. https://doi.org/10.1093/cercor/bhae331 Uddin, L. Q., Nomi, J. S., Hébert-Seropian, B., Ghaziri, J., & Boucher, O. (2017). Structure and Function of the Human Insula. Journal of Clinical Neurophysiology, 34(4), 300–306. https://doi.org/10.1097/wnp.0000000000000377
Zhang, R., Deng, H., & Xiao, X. (2024). The Insular Cortex: An Interface Between Sensation, Emotion and Cognition. Neuroscience Bulletin, 40(11), 1763–1773. https://doi.org/10.1007/s12264-024-01211-4
Acknowledgement
I want to thank my past and current science teachers who’ve taught me and helped shape my love for everything science, and my family and peers for always encouraging me to strive.