Electric Moon Dust
If humanity ever settled on the moon, what could be used for power? I am making a battery out of various metals found on both the Earth and Moon. To make my battery, I used magnesium, titanium, alluminum, iron, chromium, and D.M.M (voltage measurement).
Cyra Noori
Louis Riel School
Grade 5
Problem
If humanity ever colonized the Moon, how would we power small devices (personal life support systems, small rovers, etc.) using the available lunar resources?
My experiment will measure the difference in electric potential (voltage) between the anode and cathode of a simple battery, which uses metals found on the Moon. It will also determine which combination of electrode metals will produce the greatest voltage.
Hypothesis:
If I change the anode metal (Mg, Al, Ti, Cr) in the battery, while keeping the cathode metal (Fe) constant, then different voltages will be produced between them, because the electric potential for oxidation / reduction between the metals is different.
Background Research
Regolith exists on both the Earth and the Moon because the Earth and the Moon share a common past. We can prototype a lunar battery here on Earth by using metal elements that are found on both solar bodies. Regolith is crumbled rock covering bedrock, and it contains metals. It is derived from Greek words, rhegos (rock) and lithos (blanket).
Around four and a half billion years ago, a protoplanet around the size of Mars, called Theia, collided with our planet, the result of which formed the Moon. The blob of molten rock that came flying off the young planet was a mix of Theia and Earth, and so the Moon shares a similar regolith as our planet.
In short, we can build a basic battery with electrodes that are made from the conductive materials that are found on both the Earth and the Moon.
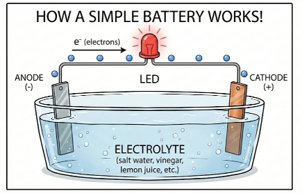
How a Battery Works
A battery uses two different metals, called the anode and cathode, sitting in a liquid solution called an electrolyte.
A chemical reaction at the anode makes it build up extra electrons, while the cathode has fewer electrons. When we connect a wire between the two electrodes, the electrons flow through the wire from the anode to the cathode. The electrolyte lets ions move inside the battery so the charges at each electrode stay balanced.
If we put a small light in the circuit, the electrons have to pass through it, and this electric current does the work which makes the light turn on.
Variables
Controlled Variables: electrolyte (type, state, temperature), cathode metal, measuring instrument (digital multimeter / DMM), alligator clips (type of metal).
Manipulated Variable: anode metal.
Responding Variable: the amount of direct current voltage produced.
Materials
● Titanium (Ti)
● Aluminum (Al)
● Magnesium (Mg)
● Iron (Fe)
● Chromium (Cr)
● Table Salt (NaCl)
● White vinegar
● Two sets of alligator clips
● Digital multimeter (DMM)
● Plastic tub

Procedure
1. Lay out the materials.
2. Make the electrolyte solution out of 500ml white vinegar & 5 tablespoons of table salt.
3. Pour the electrolyte solution in plastic tub.
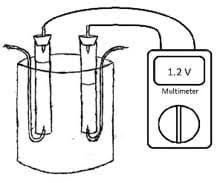
4. Put first anode metal (Cr) in left end of plastic tub.
5. Put cathode metal (Fe) in right end of plastic tub. Make sure your anode and cathode are not touching.
6. Take 1st alligator clip: attach one end to anode; attach other end to DMM.
7. Take 2nd alligator clip: attach one end to cathode; attach other end to DMM.
8. Connect one end of DMM to anode; attach other end to cathode.
9. Observe the amount of direct current (DC) voltage produced using DMM. Do three trials using the same anode.
10. Write results in your data table.
11. Repeat experiment with different anode metals (Ti\, Al\, Mg)\, keeping cathode metal (Fe) fixed.
Experiment Results / Observations
Table 1: Voltage Produced by Different Electrode Metals
| EXPERIMENT NO. |
ANODE METAL |
CATHODE METAL |
TRIAL 1 (V) |
TRIAL 2 (V) |
TRIAL 3 (V) |
AVERAGE ELECTRIC POTENTIAL / VOLTAGE (V) |
| Experiment 1 |
Chromium |
Iron |
0.290 |
0.310 |
0.310 |
0.302 |
| Experiment 2 |
Titanium |
Iron |
0.120 |
0.130 |
0.180 |
0.143 |
| Experiment 3 |
Magnesium |
Iron |
1.050 |
1.040 |
1.050 |
1.047 |
| Experiment 4 |
Aluminum |
Iron |
0.290 |
0.280 |
0.280 |
0.283 |
Analysis
During the experiments, I observed the following:
● Mg produced the highest voltage
● Using Ti as the anode resulted in the lowest voltage
● Al and Cr resulted in fairly similar voltages
● The voltage continued to decrease rapidly while the measurements were being taken
● When the Mg was taken out of the electrolyte solution, it had oxidized so quickly, that it had turned black
Aluminum Experiment

Chromium Experiment

Titanium Experiment

Magnesium Experiment

Conclusion
In the end, my hypothesis was confirmed. Using different metals as my anode resulted in different voltages being produced by my battery, even if these differences were only minimal.
Application
Applying my experiment to the future, we could be powering small lunar devices using similar, but more advanced versions of this battery.
With the natural regolith metals that are available on the Moon, it would be best to make these lunar batteries using Magnesium as the anode and Iron as the cathode, because the electric potential (voltage) that is produced between them is the greatest.
Future Experiments
If I were to do this experiment again, then I would consider changing the following:
1. Using a different electrolyte solution (e.g.\, sulphuric acid or lithium salt).
2. Changing the temperature of the electrolyte solution (hot vs cold).
3. Making a simple battery with the natural materials available on other planets (e.g.\, Mars).
4. Working on a method to prevent oxidation (rusting) of the anode metal.
I would also want to understand why the voltages started high but dropped rapidly.
Sources of Error
During my experiments, I observed that the anode metals were oxidizing as they were exposed to air. This oxidization could have decreased the electric potential.
Dissimilar masses / geometries (sizes and shapes) of the electrode metals could have impacted their respective electric potential. Ideally, we would have had standardized masses, shapes and sizes of each metal for our experiment.
Contamination of the electrolyte solution between experiments could also have potentially impacted my results.
Citations
Kegerreis, J. A., Ruiz-Bonilla, S., Eke, V. R., Massey, R. J., Sandnes, T. D., & Teodoro, L. F. A. (2022). Immediate origin of the Moon as a post-impact satellite. The Astrophysical Journal Letters, 937, L40. https://doi.org/10.3847/2041-8213/ac8d96
Korotev, R. L. (n.d.). How do we know that it is a rock from the Moon? In Some Meteorite Information. Washington University in St. Louis. Retrieved January 29, 2026, from https://sites.wustl.edu/meteoritesite/items/how-do-we-know-that-its-a-rock-from-the-moon/
Max Planck Institute for Solar System Research. (2025, November 20). Theia and Earth were neighbors. Max Planck Institute for Solar System Research. Retrieved January 29, 2026, from https://www.mps.mpg.de/theia-and-earth-were-neighbors
NASA Ames Research Center. (n.d.). New supercomputer simulation sheds light on Moon's origin [Video]. YouTube. Retrieved January 29, 2026, from https://www.youtube.com/watch?v=kRlhlCWplqk
Prysmian Group. (2024, August 19). Why use copper as a conductor? Prysmian UK. Retrieved January 29, 2026, from https://uk.prysmian.com/media/news/why-use-copper-as-an-electrical-conductor
Turner, B. (2022, October 6). Earth's Moon could have taken just hours to form from a shattered mess. ScienceAlert. Retrieved January 29, 2026, from https://www.sciencealert.com/earths-moon-could-have-taken-just-hours-to-form-from-a-shattered-mess
U.S. Department of Energy, Office of Science. (n.d.). DOE explains...batteries. U.S. Department of Energy. Retrieved January 29, 2026, from https://www.energy.gov/science/doe-explainsbatteries
Wikipedia contributors. (n.d.). Galvanic series. In Wikipedia, The Free Encyclopedia. Retrieved January 29, 2026, from https://en.wikipedia.org/wiki/Galvanic_series
Wikipedia contributors. (n.d.). Lunar resources. In Wikipedia, The Free Encyclopedia. Retrieved January 29, 2026, from https://en.wikipedia.org/wiki/Lunar_resources
Williams, M. (2015, May 28). What is lunar regolith? Universe Today. Retrieved January 29, 2026, from https://www.universetoday.com/articles/lunar-regolith
ChatGPT: I used this LLM to create the image of how a battery works and to help me research and learn more about batteries in general.
Acknowledgements
I would like to acknowledge the support and encouragement of my Grade 5 teachers at Louis Riel School. Thank you for keeping me curious!
I would also like to thank my mom and dad for helping me learn about simple batteries, how they work, and how to do a really cool experiment.
Last but not least, I would like to acknowledge the scientists that put in a lot of work and effort to help me get the answers I needed, especially Alessandro Volta!!








