Effect of Crude Okra-Derived Mucilage Concentration on Microplastic Flocculation and Sedimentation in Freshwater
Prabhneet Sidhu
Lester B. Pearson High School
Grade 11
Presentation
No video provided
Hypothesis
Testable Question:
How does increasing concentration of crude okra (Abelmoschus esculentus)-derived mucilage influence microplastic flocculation efficiency and sedimentation efficiency in a controlled freshwater system?
Hypothesis:
This study hypothesizes that as the concentration of crude okra-derived mucilage increases the microplastic flocculation efficiency and sedimentation efficiency will significantly enhance. This increase is expected due to higher polysaccharide-mediated particle-bridging interactions.
Null Hypothesis (H₀):
The concentration of crude okra (Abelmoschus esculentus)-derived mucilage has no significant effect on microplastic flocculation efficiency or sedimentation in freshwater.
Alternative Hypothesis (H₁):
Increasing concentrations of crude okra (Abelmoschus esculentus)-derived mucilage significantly enhance microplastic flocculation efficiency and sedimentation in freshwater due to polysaccharide-mediated particle-bridging interactions.
Research
Research
What are microplastics?
Microplastics are plastic particles with sizes ranging from 1 µm to 5 mm with varying morphology and chemical composition (Liu, 2025). They originate from fragments of plastic litter and environmental emissions, making them pollutants (Srinivasan et al., 2025). There are two groups microplastics can be classified into which are, primary and secondary microplastics (Srinivasan et al., 2025). Primary microplastics are manufactured for personal care use and cosmetic formulations like emulsion stabilization, viscosity regulation, synthetic clothes, etc (Srinivasan et al., 2025). These primary microplastics are introduced to the environment through discharge from industries, water treatment plants, wind deposition, or surface runoff (Srinivasan et al., 2025). Secondary microplastics are created from the fragmentation and weathering of larger plastics by wind, water, sunlight, and other forces (Srinivasan et al., 2025). Microbeads are a type of microplastic that are tiny, manufactured pieces of polyethylene plastic that are typically added to healthy and beauty products as exfoliants. Due to their tiny size, microbeads are able to easily pass through water filtration systems and into natural water sources, aquatic life, and even us (National Ocean Service, 2024). Other types of microplastics include fibers, foam, and fragments with each of their compositions vary widely (Fisheries and Oceans Canada, 2021). Microplastics most commonly found in marine ecosystems are secondary microplastics like fibres from synthetic clothes, tire duct, single-use plastics, and other sources like fishing gear and plastic bags (Fisheries and Oceans Canada, 2021). These microplastics are mainly introduced to waters from plastic litter, treated wastewater, sewer overflow from high rains, or from plastic equipment like nets (Fisheries and Oceans Canada, 2021).
Figure 1: An image demonstrating how microplastics pollute the environment and their sources Retrieved from: https://www.sciencedirect.com/science/article/pii/S0160412025001485
Why are microplastics a problem?
While the risks and dangers of microplastics on human health are still unclear, they are suspected to carry many potential risks to human and environmental health (Liu, 2025). The primary danger of microplastics comes from their behaviour as substrates for harmful contaminants to adhere onto or absorb into (Srinivasan et al., 2025). When these particles are consumed by organisms, bioaccumulation and bioamplification can occur (Srinivasan et al., 2025). This bioaccumulation can impact the organisms ability to feed, grow, move, reproduce, life expectancy, and cause inflammation (Fisheries and Oceans Canada, 2021). These impacts can be enhanced further if the plastic contains harmful chemicals such as flame retardants, colorants, and other additives that may leak into the organism. There is also a risk of exposure to other chemicals because other pollutants in water can be attracted to microplastics and stick to them (Fisheries and Oceans Canada, 2021). Microplastics pose a problem for humans are they can enter the body through inhalation and ingestion and accumulate in areas of the body like artery walls (UN Environment Programme, 2025). Microplastics also pose a problem for plants, animals, and the environment as a study has shown that they can slow the growth of marine algae (phytoplankton) which is essential for many aquatic food webs. Other studies have shown that microplastics can make soil less fertile, and potentially speed the melting of ice in places like the Arctic which in turn is speeding up global warming (UN Environment Programme, 2025). Overall, microplastics harm human, plant, animal, and environmental health, so it is critical that we find ways to mitigate and remove microplastics from our ecosystems and bodies.
Current methods/solutions used to remove microplastics from water:
How are microplastics usually removed?
Current methods and solutions for removing microplastics from wastewater use inorganic and organic polymeric flocculants that are non-biodegradeable and toxic to ecosystems (Srinivasan et al., 2025). Polyacrylamide is a synthetic flocculant commonly used in wastewater treatment systems to remove microplastics, however, under certain conditions it can break down into toxic chemicals, defeating its purpose (Tarleton State University, 2022). Other methods include filtration, coagulation, and sedimentation, each with their own strengths and limitations (Puteri et al., 2025). Methods like filtration and sedimentation sometimes fail to capture smaller, suspended microplastics due to their size and buoyancy, so coagulation and flocculation are used to aggregate and destabilize the remaining microplastics (Puteri et al., 2025). Coagulation and flocculation use charge neutralization, absorption, and bridging to neutralize the surface charge of microplastics allowing them to aggregate, and then become microflocs. The flocculation phase begins with the addition of polymers like polyacrylamides and biopolymers like chitosan which work as bridges between microflocs. These bridges between microflocs eventually create larger aggregates that either float to the surface or sink to the bottom (Puteri et al., 2025).
What are natural flocculants and polysaccharides?
In order to understand flocculants, it is important to understand flocculation first. Flocculation is a water treatment method where fine, suspended particles in water are brought together (bridging) into large clumps (flocs) by flocculants (Panalytical, 2024). Flocculants neutralize the electrical charges that keep particles apart, so the particles start to bind (bridge) and form large aggregates called flocs (Panalytical, 2024). Chemical flocculants are mainly either inorganic, organic, or composites (Porter, 2025). Some commonly used inorganic flocculants are aluminum sulfate, ferric chloride, aluminum chloride, and ferrous sulfate. Organic flocculants are typically polyelectrolytes which have multiple positive or negatively charged ions, which give them the ability to affect the surface charge of particles and create bridges between them (Porter, 2025). Polysaccharides are also considered organic flocculants, more specifically natural polymer flocculants (Zhang et al., 2023). They are biopolymers (carbohydrates) that are cationized which make them natural flocculants (Zhang et al., 2023). In comparison to conventional flocculants like polyacrylamide, a synthetic flocculant, can sometimes cause degenerative diseases whereas natural flocculants are biodegradable and biosafe (Leonhartsberger et al., 2025; Zhang et al., 2023).
Variables
Independent (Manipulated) Variable:
Final concentration of crude okra mucilage in the 15 mL reaction mixture (0%, 6.7%, 20%, 33.3% v/v).
Dependent (Responding) Variable:
Mean floc diameter (µm), suspended particle count (particles/field of view), and sedimentation efficiency (% removal).
Controlled Variables:
| Controlled Variable | Effect on Dependent Variable | How it will be controlled |
|---|---|---|
| Total Reaction Volume (15mL) | Could alter final mucilage concentration and microplastic dilution. A large volume would decrease the collision frequency between particles and polymers, potentially reducing floc formation. | All conditions were adjusted to have a total volume of 15 mL using microplastic suspension and the specified volume of okra extract. |
| Initial microplastic concentration | Higher microplastic concentrations can result in an increase in collision frequency, potentially increasing aggregation. Lower concentration can result in reduced floc formation and sedimentation efficiency. | A single stock suspension was prepared and stirred prior to being withdrawn from to ensure homogeneity across all of the trials/conditions. |
| Particle Size Range (100–500 µm) | Larger particles settle faster (Stokes’ Law). If the particle size varied significantly, sedimentation differences between trials could occur. | Ground plastic particles were sieved to isolate the particle size to about 100-500 µm. |
| Temperature (room temp, \~22°C) | The temperature affects the viscosity of the solution and the mobility of polymer chains. High temperatures could reduce the viscosity and decrease floc stability whereas lower temperature could increase the viscosity and slow settling. | All trials were done at a constant room temperature in the same indoor environment. |
| Settling Time (30 min) | As time increases, sedimentation efficiency also does, so different settling durations would directly affect the sedimentation efficiency and removal percentage. | All samples will be allowed to settle for 30 minutes undisturbed using a stopwatch for accuracy. |
| Mixing Method (10 inversions) | Insufficient mixing would reduce polymer-particle interaction whereas excessive mixing could break flocs apart and reduce the average floc diameter. | Each test tube will be inverted 10 times in 10 seconds right after the extraction is added. |
| Microscope Magnification (100×) | Different magnifications change the field size and resolution, which can affect the particle count per field and measurement accuracy. | All microscopic analysis was performed at 100× total magnification. |
| Sampling Depth (2 cm below surface) | At different depths, the particle concentration varies after settling. Sampling at different depths would create artificial variability in the measured sedimentation efficiency. | All the final samples were withdrawn consistently from 2 cm below the liquid surface using a pipette. |
Procedure
Materials
- Distilled water
- Okra pods
- Plastic material (water bottle: polyethylene terephthalate)
- Beakers (250mL)
- Graduated cylinders (10mL, 50mL, 100mL)
- Fine mesh strainer
- Smartphone camera
- Stage or ocular micrometer
- Coverslips
- Test tube rack
- Coffee filter
- coffee grinder (dedicated for lab use)
- Freezer
- Sealed containers
- Stir sticks
- Test tubes (13)
- Tube caps
- Compound Microscope
- Microscope slides
- Digital balance
- Thermometer
- Hot plate
- Pipettes (0.5mL markings)
- Stopwatch
- Masking tape and a marker for labeling
Procedure
Phase 1: Crude Okra-Mucilage Extraction
| Step in Procedure | Why/Details/Notes |
|---|---|
| Wash the okra pods with distilled water |
|
| Cut into uniform \~0.5 cm thick sections |
|
| Weigh exactly 10.00g of sliced okra using a digital balance |
|
| Add 100ml of distilled water using a graduated cylinder to a 250mL beaker |
|
| Add the okra and a thermometer to the beaker with distilled water |
|
| Heat mixture to 65°C ± 2°C on a hot plate for 30 minutes, stirring gently every 3-5 minutes |
|
| Remove from heat and allow to cool to room temperature |
|
| Filter solution through fine mesh strainer, then through the coffee filter |
|
| Measure the final volume |
|
| Store in a sealed, air-tight container with the date and time labelled at 4°C |
|
Phase 2: Microplastic Creation and Suspension
| Step in Procedure | Why/Details/Notes |
|---|---|
| Wash plastic thoroughly and dry completely |
|
| Cut into very small pieces |
|
| Freeze overnight |
|
| Grind in coffee grinder |
|
| Sieve through mesh |
|
| Weigh 0.10g of microplastics |
|
| Add microplastics to 500mL of distilled water in a beaker |
|
| Stir vigorously for 2 minutes |
|
| Determine approximate particle size range using microscope calibration |
|
Phase 3: Experiment
| Step in Procedure | Why/Details/Notes |
|---|---|
| Prepare 4 groups: control, low, medium, and high concentration |
|
| Conduct trials at constant room temperature |
|
| Label test tubes |
|
| Add okra extract to each test tube |
|
| Add microplastic suspension to each test tube |
|
| Cap tubes, then mix each by inverting 10 times over 10 seconds and start timer |
|
| Take one sample from each tube immediately after mixing (Time = 0) |
|
| Place tubes in the rack and allow to settle for 30 minutes |
|
| Record observations at; 5 min, 15min, 30min |
|
| Use a micropipette to sample 0.5mL from top (2cm deep) |
|
| Place 1 drop on microscope slide and cover with slide cover |
|
| Place onto microscope and magnify to 100x total magnification |
|
Randomly select 5 fields of view:
|
|
| Calibrate microscope using a stage micrometer |
|
| Measure and record the diameter of 15 distinct, randomly selected flocs per a condition |
|
| Capture digital images of flocs (using a phone camera) |
|
| Perform three independent replicates per condition (n = 3), for a total of 12 trials |
|
| Calculate mean +/- standard deviation | |
| Calculate sedimentation efficiency (%) |
|
Observations
Trial 1
| Time (minutes): | Controlled (0 mL) | Low (1 mL) | Medium (3 mL) | High (5 mL) |
|---|---|---|---|---|
| 0 | Uniform suspension, particles are evenly distributed | Uniform suspension, slight visible viscosity | Evenly distributed mucilage and particles, uniform suspension | Noticeable higher viscosity, uniform suspension |
| 5 | No visible settling | Even distribution of particles, very few flocs forming | Few visible flocs, somewhat even distribution of particles | Some visible flocs appear, particles are suspended towards the bottom more |
| 15 | Some settling of larger particles | More flocs forming, most particles are suspended near the middle | Particles suspended near the bottom more, more visible flocs forming | Larger flocs near the bottom, majority of particles suspended/settling to the bottom |
| 30 | Larger particles have settled, smaller particles remain suspended | Thin layer of settled particles visible | Distinct sediment layer at the bottom | Visibility settled flocs, clearer upper and middle layer |
Trial 2
| Time (minutes): | Controlled (0 mL) | Low (1 mL) | Medium (3 mL) | High (5 mL) |
|---|---|---|---|---|
| 0 | Uniform suspension, particles are evenly distributed | Increase in viscosity, particles are uniformly suspended | Uniform particle suspension, solution is more viscous | Thick appearance, uniform particle suspension |
| 5 | No change observed | Some flocs are beginning to form | Suspended particles are distributed towards the bottom more, few flocs have formed | Some visible flocs have formed, suspended particles are distributed towards the bottom |
| 15 | Larger particles start settling down | Few visible flocs appear | Some flocs are formed, some settling is observed | Some flocs have settled, more flocs have formed |
| 30 | Large particles settled, most particles are still suspended | Top to middle sections are slightly clearer, thin layer of sediment at the bottom | Solution appears clearer with flocs settled at the bottom | Solution is clearer especially towards the top, large flocs have settled |
Trial 3
| Time (minutes): | Controlled (0 mL) | Low (1 mL) | Medium (3 mL) | High (5 mL) |
|---|---|---|---|---|
| 0 | Uniform suspension, particles are evenly distributed | Particles evenly dispersed, slightly more viscous | Solution is more viscous with some strands of mucilage visible | Highest viscosity out of all conditions, uniform distribution of particles |
| 5 | No change observed | Most particles are evenly dispersed | Small flocs are visible | Some flocs are visible |
| 15 | Suspension remains uniform with some large particles settling or floating | Visible flocs begin to appear, suspended particles are closer to the bottom | Larger flocs are formed and some settling is observed | Larger flocs have formed, some sediment at the bottom |
| 30 | Large particles have settled, particles are uniformly suspended | Light sediment layer, suspended particles closer to the bottom | layer of sedimentation at the bottom, solution is clearer in the middle | Increase in overall clarity, visible layer of sediment at the bottom |
Analysis
Data
Initial Particle Count at Time 0
| Trial | Mean Count |
|---|---|
| 1 | 51.2 |
| 2 | 48.7 |
| 3 | 50.4 |
| Overall Mean | 50.1 |
Final Particle Count After 30 Minutes
| Concentration (mL) | Trial 1 | Trial 2 | Trial 3 | Mean Final | Mean % Removal |
|---|---|---|---|---|---|
| 0 (Control) | 47 | 45 | 46 | 46.0 | 8.2% |
| 1 mL | 41 | 38 | 40 | 39.7 | 20.7% |
| 3 mL | 33 | 31 | 35 | 33.0 | 34.1% |
| 5 mL | 25 | 27 | 24 | 25.3 | 49.5% |
* The removal was calculated using each trial’s initial particle count
Floc Diameter (µm)
| Concentration (mL) | Trial 1 | Trial 2 | Trial 3 | Mean Diameter |
|---|---|---|---|---|
| 0 mL | 255 | 270 | 248 | 258 |
| 1 mL | 395 | 420 | 380 | 398 |
| 3 mL | 640 | 710 | 665 | 672 |
| 5 mL | 910 | 1020 | 955 | 962 |
Standard Deviation of Final Particle Counts
| Concentration | SD (Final Count) |
|---|---|
| 0 mL | 1.0 |
| 1 mL | 1.5 |
| 3 mL | 2.0 |
| 5 mL | 1.5 |
Standard Deviation of Floc Diameter
| Concentration | SD(µm) |
|---|---|
| 0 mL | 11 |
| 1 mL | 20 |
| 3 mL | 35 |
| 5 mL | 55 |
Graphs
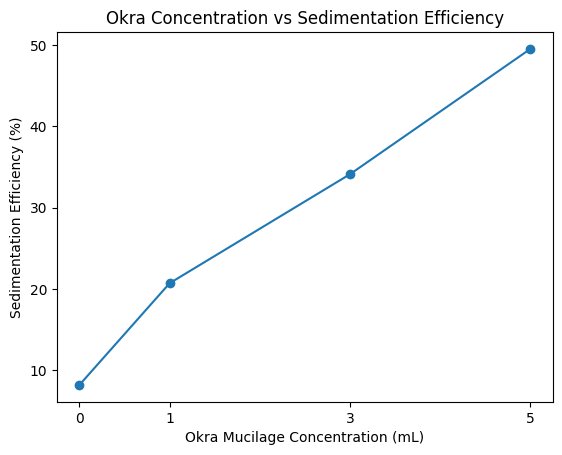
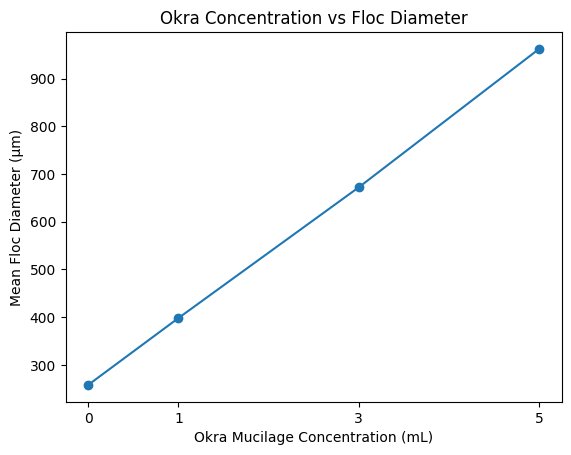
Both of the graphs demonstrate a positive relationship between the okra mucilage concentration and sedimentation efficiency (%)/floc diameter (µm). As the concentration increased the sedimentation and floc diameter also increased. This indicates a dose-dependent improvement in flocculation performance.
Conclusion
This study demonstrated that increasing concentrations of crude okra-derived mucilage significantly enhanced microplastic flocculation and sedimentation efficiency. As okra concentration increased from 0 mL to 5 mL, mean floc diameter increased nearly fourfold, and sedimentation efficiency improved from approximately 8% to nearly 50%.
The observed trend supports polymer-bridging flocculation mechanisms, in which natural polysaccharides aggregate suspended microplastics into larger flocs. According to sedimentation principles and Stokes’ Law, increased floc diameter results in greater settling velocity, which explains the corresponding improvement in removal efficiency.
The dose–response relationship indicates that okra mucilage functions as an effective biodegradable flocculant, with higher concentrations promoting stronger aggregation. While removal efficiency was moderate rather than complete, the results suggest meaningful potential for sustainable microplastic mitigation.
Given its biodegradability, low cost, and availability, okra-derived mucilage shows promise as a natural alternative to synthetic polymer flocculants currently used in wastewater treatment systems. With further optimization and scaling, this approach could contribute to environmentally sustainable engineering solutions for microplastic removal in water treatment applications.
Application
The results of this study suggest that okra-derived mucilage could potentially be used as a biodegradable flocculant for microplastic removal in wastewater treatment systems. Municipal and industrial wastewater treatment systems rely on coagulation-flocculation to aggregate suspended solids prior to sedimentation and filtration. Synthetic flocculants, such as polyacrylamide-based polymers are commonly used, however concerns exist regarding their environmental persistence and potential toxicity. A plant-derived alternative like okra mucilage offers a renewable and biodegradable option that may reduce reliance on synthetic flocculants.
By effectively increasing floc diameter and sedimentation efficiency, okra-mucilage demonstrates potential applicability in engineering clarification systems. If optimized, okra mucilage could contribute to a sustainable engineering design in water treatment systems and be incorporated into the pre-treatment stages of wastewater treatment to enhance microplastic removal before discharge.
The okra-derived mucilage could also potentially be used to mitigate microplastic pollution in freshwater systems, oceans, and in drinking water supplies. Okra is both inexpensive and widely cultivated, so its mucilage extraction is a cost-effective and biosafe solution for microplastic removal. The low-cost, plant-derived flocculant could potentially become a scalable method for microplastic removal from water in wastewater treatment systems prior to discharge, stormwater runoff filtration, and water purification systems in developing regions. Overall, use of okra-derived mucilage as an alternative to synthetic polymers and mitigating microplastic pollution aligns with sustainable engineering principles by utilizing renewable biological materials while potentially minimizing secondary environmental contamination associated with synthetic polymers.
Sources Of Error
Sources of Error
Variability in Microplastic size distribution: Particles were sieved to a range of 100-500 µm, however, variation within this range may have influenced the settling rates. The larger particles may have settled more rapidly due to increased gravitation forces, while smaller particles may have been suspended for longer. Differences in particle size distribution between trials and conditions could have affected sedimentation efficiency.
Incomplete Homogenization of Stock Suspension: Even after stirring the stock suspension prior to sampling, some microplastic particles may have still partially settled. This could result in a slight variation in initial particle concentration between trials/conditions, affecting the calculated percentage removal.
Particle Counting Error: Particle counting under the microscope was performed manually and is subject to human error like small particles being missed, overlapping particles counted as one, etc.
Environmental Temperature Variation: All trials were conducted at room temperature (approximately 22°C), however, minor fluctuations could influence solution viscosity and particle mobility, potentially altering sedimentation rates.
Further Study
Extract: A crude standardized extraction was intentionally used to evaluate real-world feasibility rather than purified polymer performance. However, the use of crude extract may introduce variability due to non-polysaccharide components, which future work could address through polymer fractionation.
Controlled Particle Size Fractions: Being able to separate the microplastic into narrower size categories would allow for an investigation of size-dependent flocculation behaviour.
Extended Settling Time Analysis: Measuring sedimentation efficiency over longer periods of time (e.g., 60 or 120 minutes) would provide insight into the long-term stability of flocs.
Comparison with Commercial Flocculants: A future study could compare the crude okra mucilage to synthetic flocculants (like polyacrylamide) to evaluate relative efficiency and sustainability.
Testing on Real Water Samples: Testing the extract in real water samples with organic matter and varying pH would provide insight into real-world performance.
Citations
Fisheries and Oceans Canada. (2021, March 29). Microplastics. Microplastics. Retrieved February 22, 2026, from https://www.dfo-mpo.gc.ca/science/environmental-environnement/microplastics-microplastiques/index-eng.html
Leonhartsberger, S., Carmona, P., Seidl, B., Mann, K.-J., Kozic, M., Sulaeva, I., Stanetty, C., & Mihovilovic, M. D. (2025, June 15). Polysaccharide-based green flocculants: A systematic and comparative study of their coagulation-flocculation efficiency. ScienceDirect. Retrieved Feburary 23, 2026, from https://www.sciencedirect.com/science/article/pii/S014486172500308X
Liu, J. (2025, November). Development and Optimization of Methods for Microplastic Analysis in Drinking Water: A Case Study of Glenmore Drinking Water Treatment Plant, Calgary. University of Alberta. Retrieved Feburary 22, 2026, from https://ualberta.scholaris.ca/items/9ac65a47-bc8c-4d62-8625-18942d5c7336
National Ocean Service. (2024, June 16). What are microplastics? NOAA's National Ocean Service. Retrieved February 22, 2026, from https://oceanservice.noaa.gov/facts/microplastics.html
Panalytical, M. (2024, September 2). What is flocculation in water treatment? Malvern Panalytical. Retrieved February 22, 2026, from https://www.malvernpanalytical.com/en/learn/knowledge-center/insights/what-is-flocculation-in-water-treatment
Porter, A. (2025). What is Flocculation and Why does it Matter in Stormwater Management. Summit. Retrieved Feburary 23, 2026, from https://summitde.com/flocculant-testing/#:\~:text=Another%20aluminum%20flocculants%20include%20aluminum,other%20hand%2C%20are%20usually%20polymers.
Puteri, M. N., Gew, L. T., Ong, H. C., & Ming, L. C. (2025, May). Technologies to eliminate microplastic from water: Current approaches and future prospects. ScienceDirect. Retrieved Februrary 22, 2026, from https://doi.org/10.1016/j.envint.2025.109397
Srinivasan, R., Bhuju, R., Chraibi, V., & Stefan, M. C. (2025, April 10). Fenugreek and Okra Polymers as Treatment Agents for the Removal of Microplastics from Water Sources. PubMed. Retrieved February 22, 2026, from https://pubmed.ncbi.nlm.nih.gov/40290963/
Srinivasan, R., Bhuju, R., Chraibi, V., & Stefan, M. C. (2025, April 10). Fenugreek and Okra Polymers as Treatment Agents for the Removal of Microplastics from Water Sources. ACS Omega. Retrieved Feburary 22, 2026, from https://doi.org/10.1021/acsomega.4c07476
Tarleton State University. (2022\, March 22). Tarleton Researchers Work to Remove Microplastics from Wastewater | Tarleton State News. Tarleton State University. Retrieved February 22\, 2026\, from https://www.tarleton.edu/news/tarleton-researchers-work-to-remove-microplastics-from-wastewater/
Tewari, A. (2022). Preparation of Microplastics for Use in Environmental Research. Tewari_Arundhati_202203_MASc_thesis. Retrieved March 2, 2026, from https://utoronto.scholaris.ca/server/api/core/bitstreams/3de50431-0f95-4e90-b55e-9a5c216124c2/content
UN Environment Programme. (2025, June 2). Everything you should know about microplastics. UNEP. Retrieved February 22, 2026, from https://www.unep.org/news-and-stories/story/everything-you-should-know-about-microplastics
Zhang, J., Li, P., Yu, Y., Xu, Y., Jia, W., & Zhao, S. (2023, September 21). A Review of Natural Polysaccharides-Based Flocculants Derived from Waste: Application Efficiency, Function Mechanism, and Development Prospects. ACS Publications. Retrieved Feburary 23, 2026, from https://pubs.acs.org/doi/10.1021/acs.iecr.3c02563#:\~:text=3.1.&text=Organic%20polymers%20are%20mostly%20used,through%20the%20adsorption%20bridging%20mechanism.
Acknowledgement
My sincerest thanks goes out to my parents for their unconditional support and limitless care no matter what. I am truly grateful for all the love and effort you put into my well-being and happiness each day. I would also like to express my utmost appreciation for my friends who constantly motivated, aided, and up-lifted me throughout my project. Thank you Rayna, Riya, and Sampannata. My special thanks goes out to Japp for truly supporting me, and for helping my project reach its highest potential. I'd additionally like to indicate my gratitude towards my mentor and coordinator, Mr. Bianchini .