The Science of pH: From Molecules to Everyday Life
Ishan Patange
Mount Royal School
Grade 7
Presentation
Hypothesis
Substances that are acidic like vinegar or lemon juice will have less pH whereas normal substances like baking soda and bleach will have more pH. The accuracy of the pH level will depend on items we use to test pH. Natural testers are cheap and ecofriendly but still can give less accurate results if other chemicals in the substance react. Whereas artificial testers give accurate results but are expensive. I predict that turmeric strip will show the best results and artificial pH strips will be accurate and better.
Research
What is pH?
pH stands for potential of Hydrogen. A substance is acidic when it has more hydrogen ions and less pH. Bases have less hydrogen ions and a more pH. The pH scale is logarithmic, meaning each number represents a tenfold change in acidity. Eg: pH 4 is 10 times more acidic than pH 5. Small numerical changes in pH can cause big chemical differences.
pH in real life
- Drinking and Waste water: pH of drinking water should be 6.5 to 8.5 for safe human consumption.
- Disadvantages of high pH drinking water: Unpleasant taste and less effect of chemicals like chlorine.
- Disadvantages of low pH drinking water: Harmful for human body and can cause damage to pipes and can also leach harmful chemicals in the entire water system.
- Swimming pools and spas: pH should be from 7.2 to 7.8.
- Cause of more pH: Chlorine becomes ineffective allowing algae and bacterias to grow.
- Cause of less pH: More acidic water and can irritate skin and other major parts.
History of pH
In 1909 a Danish chemist named Soren Peter Lauritz Sorensen introduced the concept of pH.
How is pH measured?
pH can be measured using:
- Litmus paper
- Universal indicator paper
- Digital pH meters
Factors affecting measurements of pH
- Temperature: pH meter readings depend on temperature, many meters can adapt to this change to give accurate results.
- pH depends on how hydrogen ions react. High salt concentration can affect the pH readings.
- Contamination: Dirty , old strips, and light can affect results.
Acids and Bases
Acids are substances that release hydrogen ions, have less pH. Bases are substances that release hydroxide ions, have more pH and are alkaline.
How indicators work? ( Natural)
Indicators change color when they react with hydrogen or hydroxide ions. Natural indicators contain pigments such as:
- Anthocyanins (blueberries)
- Betalains (beetroot)
- Chlorophyll (spinach)
- Curcumin (turmeric)
- These pigments change structure depending on pH, causing visible color changes.
Variables
Independent Variable:
- Type of natural pH indicator (blueberry, beetroot, spinach, turmeric, tea, coffee).
- Dependent Variable:
- Color change of the indicator strip when dipped into each liquid.
- Controlled Variable:
- Same type of paper for all strips
- Same drying time for all strip
- Same amount of test liquid in each cup
- Same lighting conditions
- Same temperature of liquids
Procedure
Step 1: Make natural pH testers.
A. pH tester out of turmeric.
1. Take 3 tsp turmeric in a small container. 2. Mix 1 cup warm water 3. Mix properly. 4. Take a coffee filter and dip it in the liquid. 5. Let it dry and use it.
B. pH strip from spinach.
1. Take 2-3 spinach leaves in a mortar. 2. Using a pestle grind it with little water. 3. Strain the liquid and dip coffee filter in it. 4. Let it dry and use.
C. pH strip from blueberries.
1. Take some frozen blueberries in a bowl. 2. Mash them with a spoon. 3. Strain the liquid and dip coffee filters. 4. Let it dry and use.
D. pH strip from beetroot.
1. Cut beetroot in to small pieces. 2. Put them in a mortar and grind it adding little water. 3. Strain the liquid and dip coffee filters. 4. Let it dry and use.
E. pH strip from tea.
1. Take boiling hot water and add tea powder in it. 2. Wait and strain the liquid. 3. Dip coffee filters in it. 4. Let it dry and use.
F. pH strip out of Coffee.
1. Take 2 tsp of water and heat it. 2. Add instant coffee in it. 3. Dip the coffee filter in it. 4. Let it dry and use.
Step 2: Get and make the indicators.
A. Get rubbing alcohol which contains 70% isopropyl alcohol. B. Get tap water. C. Get apple cider vinegar. D. Make coffee solution with water and coffee. E. Make baking soda solution with water and baking soda. F. Get concentrated lemon juice.
Step 3: Perform the experiment. A. Test with artificial pH testers and compare it with color scheme. B. Test with natural testers and classify if substance is acid, base or neutral.
Step 4: Make conclusion and compare it with hypothesis.
Observations
| Indicators | Artificial - pH strips (color pallet number.) | blueberry strip (color) | spinach strip | coffee strip | tea strip | turmeric strip | beetroot strip |
|---|---|---|---|---|---|---|---|
| Water | 7 | Purple | Stayed green | Stayed brown | Stayed light brown | No change | No change |
| Baking soda solution | 8 | Blue/ teal | Slight color change | No change | No change | reddish pink | No change |
| Apple cider vinegar | 3 | Bright Pink | No change | No change | No change | No change | No change |
| Lemon juice | 3 | Bright Pink | No change | No change | No change | No change | No change |
| Rubbing Alcohol | 5 | Magenta | No change | No change | No change | No change | No change |
| Coffee | 5 | Magenta | No change | No change | No change | No change | No change |
Analysis
Role of pigments in plants
The pigments in the fruits and vegetables for the natural pH testers played a important role reacting with the hydrogen ions in the indicators.
Blueberry contains the pigment Anthocyanins. Beetroot has betalain pigment which give it a deep red color. Spinach has chlorophyll. Turmeric has curcumin.
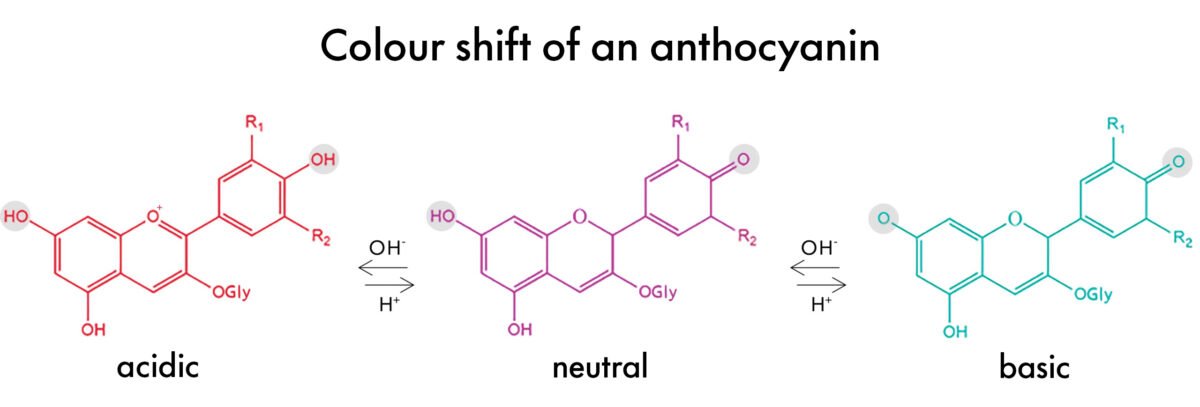
Blueberry ( Anthocyanins)
- Anthocyanins is the pigment that showed the most color change when the pH was tested.
- This is because the pigment changes its structure in acidic, neutral and basic conditions.
- Protonated structure of the pigment (flavylium cation) appears red to human eyes as it absorbs the light in a blue-green spectrum.
- Deprotonated structure appears bluish-purple to human eyes as the light is absorbed in the yellow-orange spectrum.
Beetroot (Betlain)
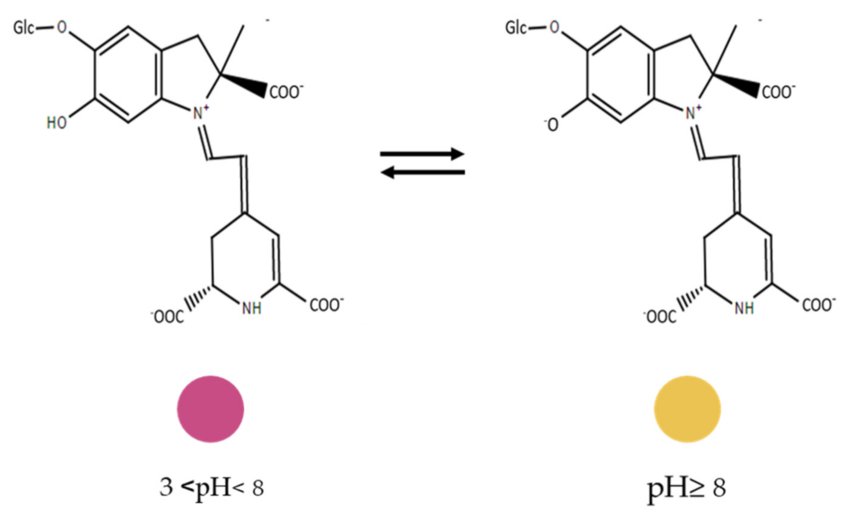
- Betalain is a water soluble pigment found in the vacuoles off the plant.
- It is pH sensitive.
- The color of the pigment in pH 3 to 7 appears is red to purple being in green region of the spectrum.
- When the pH rise the particles undergo hydrolysis and start breaking forming a pale color or sometimes brownish-yellow.
Spinach ( Chlorophyll)
- Chlorophyll mostly reacts with bases.
- In acidic conditions it looses Magnesium, forming pheophytin.
- The color changes from bright green to olive green.
- The light is absorbed in a red or blue in the spectrum.
- When the substance is alkaline magnesium retains, causing chlorophyll to degrade and form chlorophyllin.
- The color becomes more brighter green. It reflects the color green the most.
Turmeric ( Curcumin)
- Curcumin also mostly reacts with bases.
- In acidic conditions the color stays yellow, and light is absorbed in blue region in the spectrum.
- With bases the degradation increases and becomes fast. The color is reddish-brown.
Conclusion
In conclusion, artificial pH testers give accurate results because they are made in labs and in a systematic way and showed proper color changes. Blue litmus paper turns red when it detects acid. Red litmus paper turns blue when it detects deals with basic substances. Purple litmus paper can detect both alkaline and acidic substances, showing accuracy. Whereas, natural pH strips are eco-friendly but can only state if the substance is acid, alkaline, neutral. They are made with natural ingredients containing different pigments and these are affected by various factors. Fruits and vegetables can grow in different ways and these ways can affect the pigments in them. Some can have pesticides and not every fruit is grown in the same way, lacking accuracy. Among, the natural pH testers blueberry tester showed various and vivid color schemes. It is due to anthocyanins and the way they react with hydrogen ions. Hence, this shows that natural pH testers are simple for testing, but artificial pH testers are more accurate for scientific measurement.
Application
Natural pH testers are eco friendly and can be used in classrooms to educate young students about pH in a safe manner. It can also be used to check the pH of soil and other household items. Artificial pH testers are accurate and can be used to test the pH of areas that require scientific research or in labs. They have the ability to give proper changes and can be used to test water quality. Both natural and artificial pH testers are important. Natural indicators are safe and eco friendly and for basic testing, while artificial indicators provide accuracy required for scientific use, each having their pros and cons.
Sources Of Error
Natural pH testers are eco friendly and can be used in classrooms to educate young students about pH in a safe manner. It can also be used to check the pH of soil and other household items. Artificial pH testers are accurate and can be used to test the pH of areas that require scientific research or in labs. They have the ability to give proper changes and can be used to test water quality. Both natural and artificial pH testers are important. Natural indicators are safe and eco friendly and for basic testing, while artificial indicators provide accuracy required for scientific use, each having their pros and cons.
Citations
https://atlas-scientific.com/blog/why-is-ph-important/?srsltid=AfmBOoruoM96MlB9mfeWXON1GFix6WmI5Uu57odclDkq8ie6rTtDYFub Atlas Scientific. https://www.sciencebuddies.org/science-fair-projects/references/acids-bases-the-ph-scale Study buddies
https://www.youtube.com/watch?v=KfWvdSyW6Io Youtube video
https://www.britannica.com/science/pH Britannica
https://www.ndsu.edu/agriculture/sites/default/files/2022-01/fn595.pdf
https://learn.ddwcolor.com/why-do-anthocyanins-change-color/
Acknowledgement
I acknowledge :
- My mom helped me with taking video and editing it.
- Mr. Webster guided me on how to do the project.
- Thanks to school for giving me materials needed.
- My dad purchased other material needed for the experiment.

