Which common drink (Soda, Juice, milk) is more acidic (to pH indicator)
Lojin Ghazali
Peter Lougheed School
Grade 9
Presentation
Hypothesis
🐟 𓆝 ⋆。°𓆝 ⋆ What I believe might happen during the answers of my test is the soda being the most acidic, the juice being neutral, & the milk being basic. I believe that the answers will be fullt different than the other
Research
⊹₊˚‧︵‿₊୨ᰔ୧₊‿︵‧˚₊⊹
The research used for my project is about how a pH indicator strip (blue litmus paper) can be used to test whether common household liquids are most acidic, neutral, or basic (alkaline). The pH indicator changes color based on hydrogen ion concentration. The test classifies substances on the pH scale: acids have a pH less than 7, while bases have a pH greater than 7 (take a look at this image to get a clearer about the idea) !
This experiment helps in understanding the chemical nature of cleaning agents (usually basic) and food/drink (usually acidic), that's why I chose to question it once more differently.
Variables
- The type of liquid is the independent variable, while the color of the litmus paper (result) is the dependent variable
- Independent Variable (What is changed): The type of liquid being tested (milk, soda, and juice). This is the variable I am manipulating to see how it affects the litmus paper.
.✦ ݁˖
- Dependent Variable (What is measured): The color change of the blue litmus paper (whether it stays blue or turns red). I’ll measure the acidity or alkalinity of the liquid.
.✦ ݁˖
- Controlled Variables (What is kept the same): The amount of liquid I used, the amount of blue litmus paper I used, the temperature of the substances, and the duration the paper is exposed to the liquid.
Procedure
- For Preparation: Pour small amounts (10-20 mL) of milk, soda, and juice into separate 3 glasses
- For testing my question Dipping a new strip of blue litmus paper into each sample will do.
- For Replication: A red litmus paper to confirm my results (bases turn red paper blue).
Observations
The observations I took during the experiment was....
- Juice (Vimto): Blue litmus paper turns like a redish pink, indicating it is acidic due to citric or tartaric acids.𝄞⨾𓍢ִ໋
- Milk: on blue litmus paper turns slightly purplish, because milk is slightly acidic (pH around 6.5–6.7), which makes it almost neutral. 𝄞⨾𓍢ִ໋
- soda (Pepsi): Due to carbonic acid, it often turns blue litmus red or light pink.𝄞⨾𓍢ִ໋
Analysis
Where things could've been wrong with my experiment:
wrong measurements of drinks: ex, milk being too much more than soda different containers and shapes for the seperate drinks might cause confusion, creating unoticable errors.
Conclusion
The experiment concludes that soda and juice are most acidic, while milk is generally neutral to slightly acidic. Soda and juices turn blue litmus paper red due to low pH. Milk typically shows no change or a slight shift, indicating a neutral or near-neutral pH (around 6.5–7).
My hypothesis was somewhat correct since the soda is the most acidic but so was the juice in which I got wrong thinking it'd be fully different, milk was also found to be on the neutral side in which I thought was going to be on the basic side.
The experiment confirms that sodas and juices are acidic (pH < 7), while milk is close to neutral or slightly acidic! Now you know how truly acidic your eveyday common drinks are!
Application
The application of testing milk, soda, and juice with blue litmus paper is to classify household liquids as acidic, neutral or basic (alkaline) based on pH. Blue litmus turns red in acids (juice, soda) and remains blue in bases (some milk, baking soda solution), demonstrating foundational acid-base chemistry.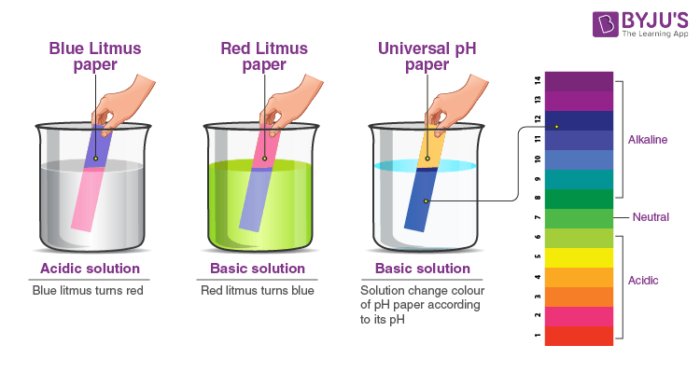
Sources Of Error
Thankfully there was no errors or remakes due to mistakes during the proccess of the experiment. only an incorrect thought, I thought that the liquids will change color but it's only the pH indicator that changes.
Citations
Best resources (websites) that helped me think & shape my ideas together:
https://www.scienceequip.com.au/blogs/news/litmus-paper-everything-you-need-to-know?srsltid=AfmBOoriljBLM8xp2y5cnSDkD2UlRl5OZ9qSLP0fb29bhRlADZE0a2Mf#:\~:text=The%20litmus%20paper%20test%20seems,which%20causes%20the%20colour%20change.
https://www.youtube.com/watch?v=3BfxiSJ5-hY
https://learning-center.homesciencetools.com/article/acid-base-reactions-ph-experiments/#:\~:text=Litmus%20is%20a%20natural%20acid,%3E%3E
https://www.sciencebuddies.org/science-fair-projects/science-projects/ninth-grade
and of course the many credits to my teachers and my parent.
Acknowledgement
₊˚ ‿︵‿︵‿︵୨୧ · · 🌊🌀🌊 · · ୨୧‿︵‿︵‿︵ ˚₊ I wanna say thank YOU to all the PLS's lovely Science fair teachers — Ms. Olfert, Ms. Duncan, Ms. Nanji, & Ms. Kim! ⊹ ࣪ ﹏﹏𓂁﹏⊹ ࣪ ˖
- Not to mention the help of my dearest mother, who has got my hopes back right after I gave up when I found out that the fishtank & the fish craft sticks idea was impossible in such short time!
₊˚ ⠀⠀⠀⠀⠀⠀⠀⠀⣾⣛⡿⡿⢽⣶⣤⣀⣀⣀⣀⣀⣀⣠⣴⣤⣄⣀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀ ⠀⠀⠀⠀⠀⠀⠀⢠⣿⢬⡭⢭⣭⣽⣗⡋⠈⠉⣿⣿⣿⣿⣿⣿⣿⣿⣷⣦⣀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀ ⠀⠀⠀⠀⠀⠀⠀⡞⠁⣂⡠⠾⢛⣛⣀⣀⠀⠀⠙⠛⢿⣿⣿⣿⣿⣿⣿⣿⣿⣿⣶⣤⣤⣄⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀ ⠀⠀⠀⠀⠀⠀⠀⣟⠛⠃⡀⣀⡉⠀⠄⠄⠈⠉⠑⠢⣄⡉⠻⣿⣿⣿⣿⣿⣿⣿⣿⣿⣿⣿⣷⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀ ⠀⠀⠀⠀⠀⠀⠀⠈⠉⠉⠁⠀⠀⠀⢀⡀⠀⠀⠀⠀⠀⠈⠲⣍⣿⣿⣿⣿⣿⣿⣿⡉⠛⢿⣿⣷⣄⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀ ⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⣰⢿⣿⣖⠿⣯⢆⠀⠀⢁⠞⠉⠙⠻⠿⣿⣿⣿⣿⣦⣀⣩⣿⣿⣷⡄⠀⠀⠀⠀⠀⠀⠀⠀ ⠀⠀⠀⠀⠀⣤⡄⠀⠀⠀⢀⡴⠛⢓⡜⢿⣿⣆⢀⢿⠀⠀⢻⣄⡀⠀⠀⠀⢒⣛⣫⣿⡟⠻⢿⠿⣾⡻⣿⡆⠀⠀⠀⠀⠀⠀⠀ ⢀⡀⢤⣤⢾⣿⣷⠄⢀⡠⠈⣄⠤⠀⠛⢾⣿⣿⣾⡎⠀⠀⠀⠈⠛⠒⠀⠀⠀⠀⠀⢈⣑⡒⠤⠄⣀⣈⣭⠗⠀⠀⠀⠀⠀⠀⠀ ⡏⠈⠉⠉⣿⣿⣿⠍⠹⣿⡄⠉⠻⢶⣌⠀⠻⣿⣿⣿⣷⣶⣴⣾⣿⣿⣿⣿⣿⣿⣶⣶⣶⣶⣶⣦⣄⣒⣊⠀⠀⠀⠀⠀⠀⠀⠀ ⠹⡦⡁⠀⠸⡟⣿⣤⢬⢿⠀⠀⠀⠀⠉⡷⣄⠙⠻⣿⣿⣿⣿⣿⣿⣿⣿⣿⣿⣿⣿⣿⣿⣿⣿⠿⣿⣿⣿⣿⣿⣶⣤⣤⣀⠀⠀ ⠀⠙⢾⡴⠄⡀⢸⣿⡏⠁⠀⠀⠀⠀⠀⠈⠪⡓⢤⡈⠙⠻⢿⣿⣿⣿⣿⣿⣿⣿⣿⣿⣿⣷⣷⣿⣟⢵⣆⣈⣍⣿⣿⣿⣿⣷⡄ ⠀⠀⠀⢷⣿⣿⣿⣿⣧⡀⠀⠀⠀⠀⠀⠀⠀⠈⠂⢌⡓⠢⢤⣉⠉⠛⠻⢿⣿⣿⣿⣿⣿⡿⠿⣿⣿⣿⠿⠛⠛⠛⠛⢛⣨⣿⡗ ⠀⠀⠀⢰⣿⣿⣿⣿⣿⣿⣦⣀⠀⠀⠀⠀⠀⠀⠀⠀⠈⠑⠲⠤⠍⣒⡂⠀⢼⡉⠛⠁⠀⠀⠀⣠⠟⠻⠙⠉⠀⠁⣉⣁⡠⠔⠀ ⠀⠀⠀⠸⣿⣿⣿⣿⣿⣿⣿⣿⣷⣤⡀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠈⠁⠒⡇⠀⠀⠀⠀⠈⢀⡠⠔⠒⠀⠉⠉⠀⠀⠀⠀⠀ ⠀⠀⠀⠀⢹⡘⢿⣿⣿⣿⣿⣿⣿⣿⣿⣶⣿⡿⣕⢆⠀⠀⠀⠀⠀⠀⠀⠀⠀⠳⠤⠄⠰⠤⠚⠁⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀ ⠀⠀⠀⠀⠀⢳⡎⣿⣿⣿⣿⣿⣿⣿⣿⣿⣿⣿⣶⣺⡄⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀ ⠀⠀⠀⠀⠀⠀⢻⣿⣾⣿⣿⣿⣿⣿⣿⠿⢿⣿⣿⣿⠁⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀ ⠀⠀⠀⠀⢀⡶⠏⠉⠻⢿⣿⣿⣿⣿⣿⡷⠆⠙⠿⣿⣧⡀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀ ⠀⠀⠀⠀⠘⣄⠀⠀⠀⠈⣛⣿⡿⠟⢿⣷⣮⣸⡐⠛⣿⣷⡀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀ ⠀⠀⠀⠀⠀⠙⠯⢒⣲⠄⠀⠀⠀⢄⣠⠀⠈⠿⣿⡀⠀⢻⣿⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀ ⠀⠀⠀⠀⠀⠀⠀⠀⠈⠉⠉⠉⠐⠲⢌⣓⠤⣀⠀⠀⠀⠈⢳⠄⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀ ⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠈⠑⠂⠭⠝⣒⠻⠟⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀⠀

